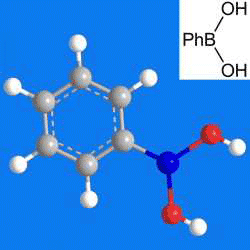
Phenylboronic acid, or benzeneboronic acid, is prepared by the reaction of the Grignard reagent PhMgBr with B(OMe)3. It and its aryl-substituted derivatives are commonly used as building blocks in synthetic organic chemistry—most often in the Suzuki coupling reaction, in which a boronic acid reacts with an organic halide to form a carbon–carbon bond. Boronic acids’ reactions with 1,2- and 1,3-diols are useful for saccharide recognition.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

