What molecule am I?
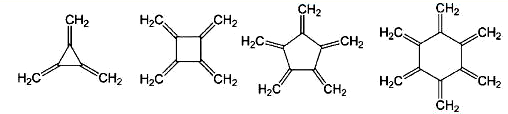
Radialenes are highly symmetric polyolefins that are difficult to synthesize and are studied mostly to characterize their bonding. They consist of regular carbon-atom polygons that are completely substituted with exo-double bonds.
The four simplest radialenes, with three-, four-, five-, and six-membered rings, are shown here. The synthesis of [5]radialene was reported only last October by Michael N. Padden-Row, Michael S. Sherburn, and colleagues at Australian National University (Canberra) and the University of New South Wales (Sydney).
The syntheses of [3]- and [4]radialene were accomplished much earlier (1965 and 1962, respectively). Four research groups prepared [6]radialene in the 1970s. All of these radialenes are unstable toward polymerization unless they are stored at dry ice temperatures.
Many theoretical studies have investigated the bonding of these and higher radialenes. Derivatives of the lower radialenes have been synthesized and their bond lengths and planarity measured. These studies show unusually long ring bond lengths in the planar [3]- and [4]radialenes and normal ring bond lengths in the twist-boat conformation of [6]radialene.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

