
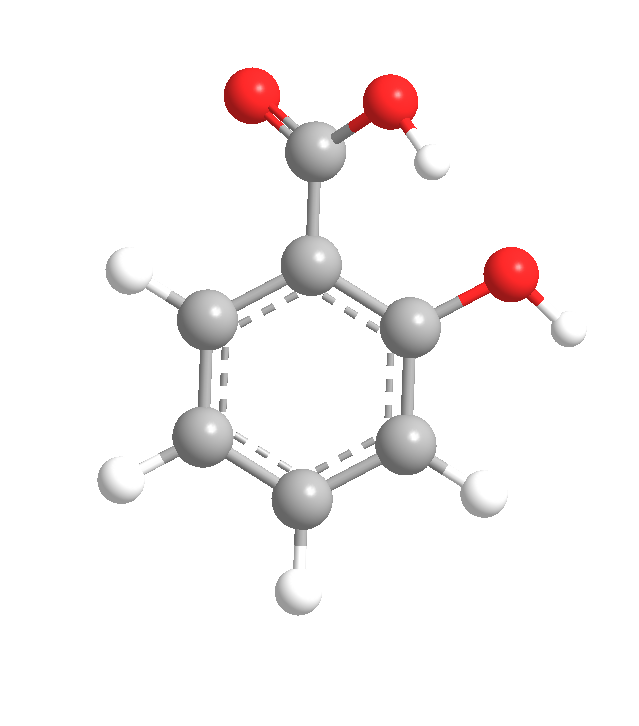
Salicylic acid (2-hydroxybenzoic acid) is a white solid first isolated from the bark of willow trees (Salix spp.), from which it gets its name. It also occurs as the free acid or its esters in many plant species.
In an early (1966) biosynthetic process, researchers at Kerr-McGee Oil Industries (now part of Andarko Petroleum) prepared salicylic acid via the microbial degradation of naphthalene. It is now commercially biosynthesized from phenylalanine.
Acetylsalicylic acid (aspirin), a prodrug to salicylic acid, is made by an entirely different process. Curiously, salicylic acid is also a metabolite of aspirin.
Salicylic acid and its esters are used as food preservatives, in skin-care products and other cosmetics, and in topical medicines. In 2015, J. L. Dangl, S. L. Lebeis, and co-workers at the University of North Carolina, Chapel Hill, discovered that native salicylic acid plays a role in determining which microbes are in the root microbiome of Arabidopsis thaliana, a weed that grows in Europe and Asia.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

