What molecule am I?
Take a look at the list of ingredients on the label of your favorite body wash, face wash, or shampoo. More than likely, sodium laureth sulfate is the first ingredient (after water) on the list.
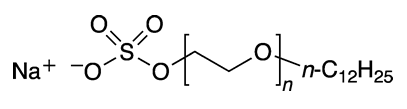
But wait—“laureth” isn’t a chemical term, is it? Well, it is, sort of. It’s a contraction for “lauryl ether” in its more formal name, sodium lauryl ether sulfate, or SLES.
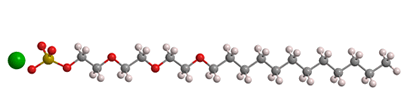
Lauryl, of course, is an early term for what is now called the dodecyl alkyl group, or n-C12H25. Ether refers to one or more ethylene oxide groups placed between the sulfate and lauryl moieties. So SLES is an oligomer that contains a few ethylene oxide linkages; the mean quantity is about 3, as shown in the 3-D image.
SLES is the most common of a group of commercial sodium and ammonium alkyl and alkyl ether sulfates. It is an inexpensive surfactant that foams easily and is an excellent detergent. It’s essentially a synthetic soap.
The commercial product is an aqueous solution that contains 30–70 wt% of SLES. Its properties are similar to those of its non-ether cousin, sodium lauryl sulfate (SLS). Both are used extensively in household and industrial cleaners, as well as in personal care products.
In the past few years, several postings on the Internet claimed that SLES and SLS can cause cancer. This myth has been debunked by Snopes.com and other Web sites. The surfactants’ worst adverse effect is skin irritation; and they should certainly be kept away from the eyes.
Sodium laureth sulfate
fast facts
| CAS Reg. No. | 9004-82-4 |
| Molar mass | 420.54 g/mol (n =3) |
| Formula | C18H37NaO7S (n =3) |
| Appearance | Clear to slightly hazy solution |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

