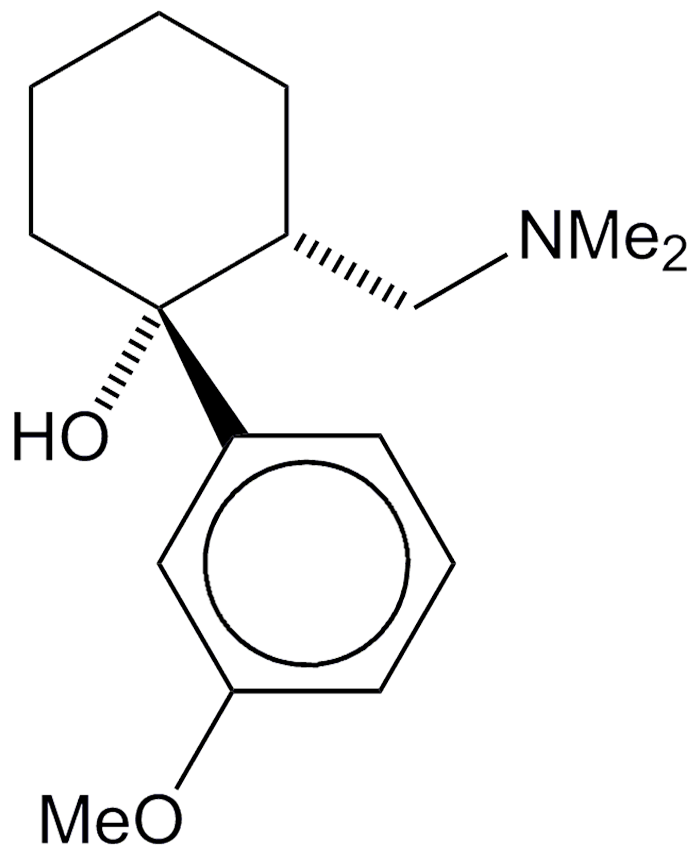
Tramadol is a venerable opioid analgesic. Its preparation was first reported in 1965 in a British patent to the West German company Grŭnenthal GmbH. It was not until 1977 that Grŭnenthal marketed the drug under the trade name Tramal. Twenty years later, companies in the United States, the United Kingdom, and Australia began to sell tramadol under various names.
Commercial tramadol consists primarily of two of its four stereoisomers, (R,R) and (S,S). (R,R)-tramadol is shown in the images. It has at least six known mechanisms of action, including as a μ-opioid receptor agonist and as a serotonin releasing agent. It has serious side effects, including seizures and risk of suicide.
Tramadol is a synthetic product that was designed to contain the structural portion of morphine that gives it its pain-killing activity. But in 2013, M. De Waard and several colleagues in France, Cameroon, and Switzerland reported its occurrence in the Cameroonian pincushion tree (Nauclea latifolia). Although De Waard’s experiments took pains to rule out contamination of the plants by synthetic tramadol, in September 2014, however, a study by M. Spiteller and coauthors in Germany and Cameroon showed that tramadol in the trees is the result of human contamination, most likely because natives in northern Cameroon administer the drug to working farm animals so that they tire less easily. The authors believe that the animals’ urine contaminates the soil where the trees grow.
Two other scientists in the field are not convinced by either research team’s argument. All of the parties are conducting additional studies to resolve the question. The answer may depend on whether the isotope ratios and stereoisomer distribution of synthetic and “natural” tramadol match up.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

