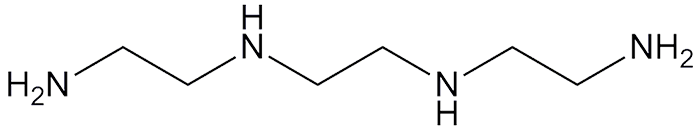
The linear polyamine triethylenetetramine is a high–boiling point, oily liquid that was first prepared in 1890 by A. W. von Hoffmann. He built on the synthesis of ethylenediamine by using condensation reactions between chloramines. Today, triethylenetetramine is made by heating ethylenediamine or ethanolamine with ammonia and then separating it from the product mixture.
Triethylenetetramine’s primary commercial use is curing epoxy resins. It is also used in analytical procedures for determining nickel and copper; it is a lubricating oil additive; and in medicine, along with penicillamine, it is used to remove copper from the body as a treatment for Wilson’s disease.
In 2009, P. Anzenbacher, Jr., and M. A. Palacios at Bowling Green State University (OH) used triethylenetetramine as a reagent in “attoreactors” made from electrospun polyurethane nanofibers. (An attoliter [aL] is 10–15liters.) Combining fibers doped with the tetramine with others doped with dansyl chloride and heating the fiber structure produced fluorescent dansylamides on the zeptomole scale (10–21 mol). A 5-aL reactor contains only ≈1500 molecules!

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

