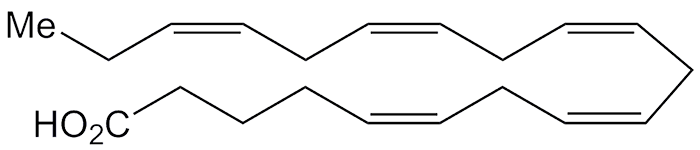
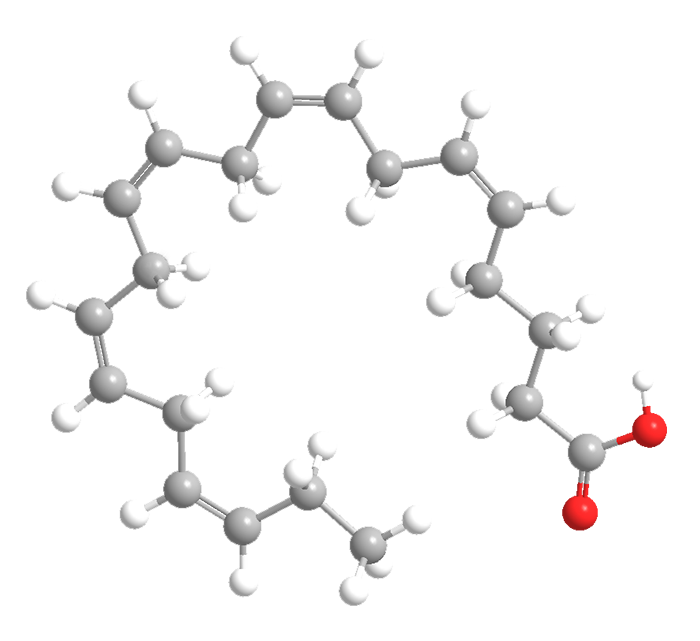
5,8,11,14,17-Eicosapentaenoic acid (EPA) is a straight-chain polyunsaturated fatty acid whose double bonds all have the cis configuration. E. Klenk and D. Eberhagen isolated EPA from cod-liver oil in 1957. It is a biochemical precursor to the prostaglandin-3, thromboxane-3, and leukotriene-5 lipid families.
The supply of marine fish as the main source of EPA and other so-called omega-3 fatty acids is limited and unsustainable. Recently, Q. Zhu and co-workers at DuPont engineered Yarrowia lipolytica, an oleaginous yeast that accumulates lipids, to produce large amounts of EPA. These researchers believe that other Y. lipolytica strains may be able to produce other fatty acids and lipids for uses such as biodiesel fuel.
MOTW update:
July 02, 2018
An eicosapentaenoic acid (EPA) isomer was the Molecule of the Week for September 30, 2013. It’s a fish oil–derived omega-3 fatty acid, which at that time was being synthesized by engineered bacteria. But there is now a method for producing it from another natural source: krill oil. During purification, EPA is converted to its ethyl ester and then restored to its free acid form.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

