What molecule am I?
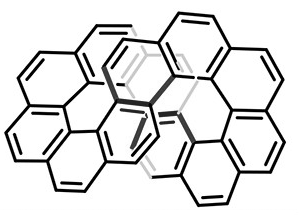
Infinitene is a chain of 12 fused benzene rings that forms an infinity symbol (∞) or, rotated 90°, a figure 8. After years of anticipation, it was synthesized in 2021 by Maciej Krzeszewski, Hideto Ito*, and Kenichiro Itami* at Nagoya University (Japan). Its official chemical name is in the fast facts table; but it is also known by the daunting term cyclo[c.c.c.c.c.c.e.e.e.e.e.e]dodecakisbenzene.
The structure of infinitene can also be thought of as two fused [6]helicene units. [6]Helicene is, of course, helical, and thus exists in two enantiomeric isomers with opposite chirality. To complete the infinitene loop, both helicenes must be of the same chirality, so that infinitene also exists in two enantiomers, designated as (+)-(P,P) and (–)-(M,M). The axial chirality symbols (P) and (M) stand for “plus”, or “right-handed”, and “minus”, or “left-handed”, respectively.
After synthesizing infinitene, Krzeszewski et al. performed all the necessary spectroscopic studies to confirm its structure. They also calculated the aromaticity of each benzene ring; but they did not report whether the entire molecule is aromatic. Subsequently, Mesías Orozco-Ic*, Rashid R. Valiev, and Dage Sundholm* at the University of Helsinki used sophisticated calculations to show that the molecule is aromatic; when an external magnetic field is applied, it creates two non-intersecting current density pathways along the molecules’ edges.
The synthesis of infinitene generated considerable buzz among chemists. C&EN readers, as well as other groups, voted it molecule of the year for 2021.
Molecules from the journals
Psychotridine1,2 is a complex alkaloid found in several of the more than 1500 species of tropical plants in the genus Psychotria, notably the Brazilian species P. colorata. Psychotridine is a probable N-methyl-D-aspartate receptor antagonist that has shown analgesic and antifungal properties and cytotoxicity against human cancer cell lines. This March, Tony Z. Scott, Vinicius F. Armelin, and Mohammad Mascagni* at MIT (Cambridge, MA) reported the first enantioselective total synthesis and stereochemical assignment of (−)-psychotridine.
[2.2.2]Cryptand3 is a widely used chelating agent for alkali metal cations. In the late 1960s, Jean-Marie Lehn at the University of Strasbourg (France) synthesized it and other cryptands; for this work, he shared the 1987 Nobel Prize in Chemistry. In March, Zhong-Ming Sun and collaborators at Nankai University (Tianjin, China) and Regensburg University (Germany) reported the first synthesis of a cyclophosphorus analogue of ferrocene; they used [2.2.2]cryptand-chelated potassium ion as the counterion for preparing the anion [Fe(P4)2]2–.
1. CAS Reg. No. 52617-25-1.
2. Not to be confused with psychotrine, CAS Reg. No. 7633-29-6., a component of ipecac.
3. CAS Reg. No. 23978-09-8.
Molecules from the Journals
MOTW briefly describes noteworthy molecules that appeared in recent ACS journal articles. See this week's
edition below.
Infinitene fast facts
| CAS Reg. No. | Racemic: 2744250-32-4 (P,P): 2744371-11-5 (M,M): 2744371-12-6 |
| SciFinder nomenclature | 10,21,9,22-[1,3,5]Hexatriene[1,3,4,6] tetrayldiphenanthro[3,4-c:3′,4′-l]chrysene |
| Empirical formula | C48H24 |
| Molar mass | 600.70 g/mol |
| Appearance | Yellow solid with green fluorescence |
| Melting point | Not reported |
| Water solubility | Insoluble |
MOTW update
Oxybenzone was the Molecule of the Week for July 16, 2018. It is a long-time sunscreen ingredient that in recent years was found to damage coral reefs. Beginning in 2021, sunscreens containing oxybenzone were outlawed in Hawaii.
William A. Mitch and colleagues at Stanford University (CA) have now discovered how oxybenzone damages coral. When coral metabolizes the compound, it converts its hydroxyl group to a glucoside. In contrast to the sunblocker oxybenzone, the metabolite is a phototoxin; when it absorbs sunlight, it forms reactive oxygen species that corrode coral tissue.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

