
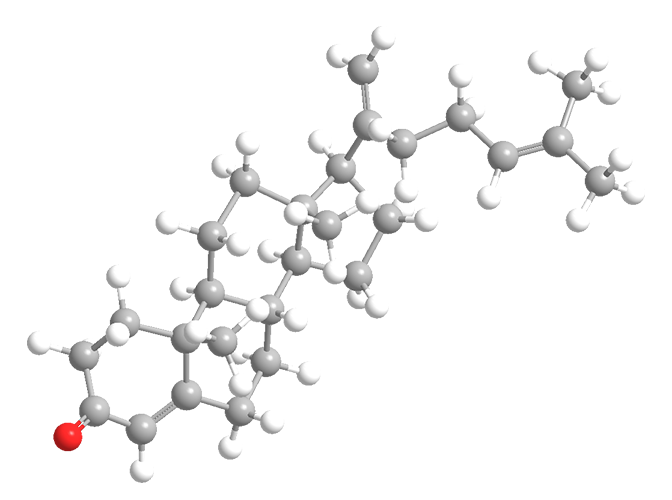
Pentalinonsterol is a steroid that was recently isolated from the Mexican and Central American pentalinon plant (Pentalinon spp.). Its structure is sort of an oxidized version of cholesterol.
Mayan healers in the Mexican state of Campeche used pentalinon roots to treat cutaneous leishmaniasis. Curious about the ingredient in the roots that cleared up the skin sores, Abhay R. Satoskar and co-workers at Ohio State University (Columbus) extracted 20 compounds from the roots and tested them against the disease. They found a “hit” in pentalinonsterol.
To obtain enough pentalinonsterol for determining whether it is a drug candidate, root extraction is not the answer. Instead, the researchers synthesized the compound from the hormone pregnenolone in five steps.
When Satoskar et al. treated mice with pentalinonsterol, they found that it is potent against visceral leishmaniasis. This disease, caused by the protozoan Leishmania donovani, is difficult to treat; many current drugs have toxic side effects, and the parasite has become resistant to many others. Satoskar’s group is now working to formulate pentalinonsterol and to identify its molecular target.
MOTW Update
In the December 7, 2015, Molecule of the Week, we reported that riboflavin is an efficient E-to-Z olefin photocatalyst. Now the same researchers, J. B. Metternich and R. Gilmour at the University of Munster (Germany) report that riboflavin can also catalyze ring-closing C–O bond formation reactions. In an example of using the vitamin to effect both reactions in tandem, the researchers converted (E)-cinnamic acids first to the Z-isomers and then to coumarins.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

