What molecule am I?
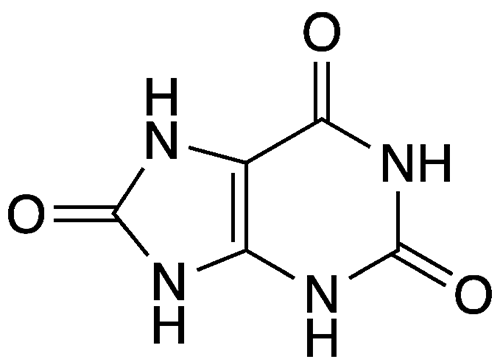
Uric acid is a purine derivative and an oxidative metabolic product of purine nucleotides in humans and other carnivorous animals. It is a weak acid with a pKa of 5.6 and is sparingly soluble in water.
In 1776, Swedish chemist Carl Wilhelm Scheele discovered uric acid in human urine and kidney stones. The same year, his discovery was confirmed by his colleague Torbern B. Bergman. Uric acid is also found in the feces of birds, reptiles, and some mammals.
Ukrainian–Austrian chemist Ivan Horbaczewski reported an early synthesis of uric acid from glycine in 1882. More recently (1962), C. Wayne Bills and co-workers at the University of Colorado (Boulder) serendipitously synthesized uric acid from urea while they were seeking a new route to pyrimidines.
Uric acid is notorious for the excruciating pain it produces in gout patients. When the acid is present in high concentrations in the blood, its low solubility causes it to precipitate as sharp crystals in and around joints, notably in the feet. Uric acid concentration increases in the body when people consume large amounts of purine-rich foods such as red meat and seafood.
There has been some speculation that the presence of uric acid and its concomitant gout play a role in COVID-19, but this connection would be indirect at most. People with gout tend to have other serious health problems such as obesity, kidney disease, and cardiovascular disease, which uric acid can exacerbate. According to Theodore R. Fields at the Hospital for Special Surgery (New York City), “Gout does not typically affect immune system function.” Adds Shailendra Singh at White River Medical Center (Batesville, AR), “People with gout don’t have a greater risk of getting coronavirus than the general population, but they do have an increased risk of complications if they do get it.”
Uric acid hazard information
| Hazard class* | Hazard statement |
|---|---|
| Not a hazardous substance or mixture |
* Globally Harmonized System of Classification and Labeling of Chemicals.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Uric acid fast facts
| CAS Reg. No. | 69-93-2 |
| SciFinder nomenclature | 1H-Purine-2,6,8(3H)-trione, 7,9-dihydro- |
| Empirical formula | C5H4N4O3 |
| Molar mass | 168.11 g/mol |
| Appearance | White crystals or powder |
| Boiling point | 310 ºC (dec.) |
| Water solubility | 60 mg/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

