During World War II, Vesta Stoudt, who had two sons serving in the U.S. Navy, was working at the Green River Ordnance Plant near Amboy, Illinois.
She helped pack boxes of ammunition. This involved waterproofing the boxes by sealing them with paper tape and dipping them in wax. A loose end of the tape was left sticking out so soldiers could pull on it and quickly peel off the whole strip of tape to open the package. But there was a critical problem with this technique. It succeeded in waterproofing the boxes, but the flimsy tabs of paper tape often tore off, leaving the boxes sealed shut.
Under enemy fire, soldiers would have had little time to deal with a hard-to-open box. They needed to unseal ammunition boxes quickly. So, Stoudt came up with a solution. She designed a waterproof cloth tape that was sturdier than the paper tape the army had been using. She proposed the idea to her bosses, but they didn’t show much interest.
Undeterred, Stoudt wrote a letter to then-President Franklin D. Roosevelt on Feb. 10, 1943. Roosevelt saw the merits of her idea and instructed the War Production Board to follow up. The Industrial Tape Corporation won the job of producing the tape in large amounts. Thus, duck tape—now known as duct tape—was invented.
Duct tape has now been used to fix an amazing variety of objects not only in the military but also for everyday repairs. It has even been critical during space missions.
Stoudt’s story illustrates many things, not the least of which is that necessity is the mother of invention. It also highlights the importance of overlooked inventions that have become so common that we forget that they needed inventing in the first place. Adhesives, of which duct tape is just one type, fall into this category. Not only have they helped solve critical, short-term problems such as packaging munitions, they’ve played crucial roles in the evolution of human culture—and sticky innovations will surely help us tackle problems that seem unsolvable today.
The original hot glue
Long before we started using synthetic tapes for packaging and mending ripped books, the advantages of stickiness had to be recognized and harnessed. Archaeological evidence suggests that glue goes back to the early-human and Neanderthal days. Starting as far back as 200,000 years ago during the Paleolithic Period, glue was likely made by heating birch bark to make a sticky tar. Tar is a natural polymer—that is, it consists of a large number of similar molecules (monomers) bonded together in long chains. It’s also thermoplastic, which means it softens when heated and solidifies as it cools. At room temperature, tar is solid, but warm it up to 29 °C (85 °F), and you can mold it like putty. When cooled, the tar hardens and can hold two things together. It can also be heated again for reuse.
Archaeologists have found evidence that tar was used for hafting—that is, attaching stone blades to wooden handles—which represented a toolmaking advancement for early humans.
Another important development in the history of adhesives was the use of the naturally occurring rock limestone (calcium carbonate, CaCO3), which is often composed of the skeletal remains of marine organisms. When heated, added to water, and mixed with sand, limestone produces lime mortar. When lime mortar reacts with carbon dioxide (CO2) in the air, it hardens into limestone, binding together the materials it’s in contact with. Lime mortar was used to bond together stone in ancient structures, including the Egyptian pyramids and the Great Wall of China.
Additives, specifically organic, protein-like materials such as ox blood were sometimes mixed into the lime mortar to enhance its performance.
Early adhesives based on animal or plant materials were strong, and many examples of structures built with these natural glues are still standing today. But ultimately, advances in technology, medicine, and culture created opportunities to invent more versatile modern adhesives, such as surgical, electrical, and gift-wrapping tapes.
Sticking with Limestone
Limestone has played a key role in the construction of some of the world’s most iconic structures. The process of making it is a cycle of the following reactions:
It starts with heating solid limestone, CaCO3, to form calcium oxide, CaO.
CaCO3 (s) + heat → CaO(s) + CO2(g)
Water is added to form lime putty, or calcium hydroxide, Ca(OH)2.
H2O(l) + CaO(s) → Ca(OH)2(s)
Over time the calcium hydroxide reacts with CO2 from the air to harden into limestone.
Ca(OH)2(s) + CO2(g) → CaCO3(s) + H2O(g)
Synthetic stickiness
Today, manufacturers don’t have to heat birch or collect ox blood to make glue or adhesive backing on tapes. Instead, they process tacky materials in manufacturing plants.
Acrylate-based polymers are commonly used to make sticky materials. Similar to all polymers, acrylate polymers are large molecules consisting of many identical, repeating units. Producing polymers makes use of something called a polymerization reaction—that is, a large number of identical molecules, called monomers, react to form one single, large molecule, the polymer. Poly(n-butyl acrylate) is a typical acrylate polymer used in adhesives. It’s made by polymerizing the monomer n-butyl acrylate.
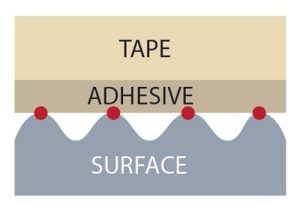
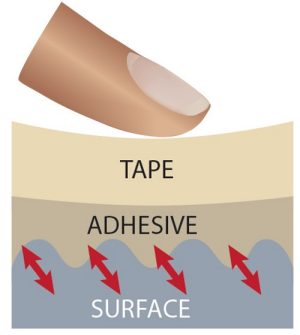
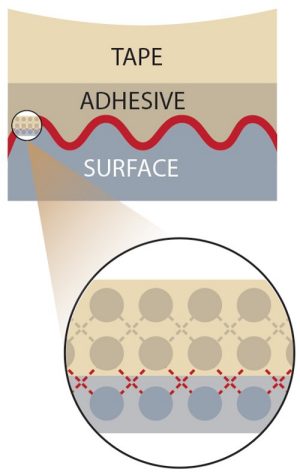
These polymers are often the sticky component in tape. The principle of how tapes work appears straightforward: Many tapes that you might use, such as transparent Scotch tape, consist of a backing material that is coated on at least one side with a layer of a pressure-sensitive adhesive. It’s pressure sensitive because you have to apply a light force to make it stick effectively.
Behind this apparently simple mechanism, there is a lot of science involved. Zoom in on the molecular level, and you’ll find that tape stickiness relies on the balance between adhesive and cohesive forces. Adhesive forces are attractions between molecules in different substances. Cohesive forces are attractions between molecules in the same substance. To better understand these two forces, consider a drop of rain on a car windshield. Adhesive forces between the water molecules and the glass hold the water in place. Cohesive forces occur between water molecules and hold them together in a droplet. Without cohesive forces, the water molecules would spread out on the glass.
Here’s how these forces work in tape. In many tapes, adhesive forces involve van der Waals forces. Van der Waals forces are attractive interactions at the atomic and molecular level, but are much weaker than covalent and ionic bonds. In order for tape to stick to a surface, van der Waals forces must be established between the molecules in a piece of tape and the molecules or atoms at the surface. Because van der Waals forces are distance-dependent and vanish as the distance between molecules increases, applying a light force on a piece of tape with your finger pushes the adhesive and surface molecules close enough together to establish van der Waals forces and cause the tape to stick.
Picture it this way: Let’s say you’re taping a poster to your bedroom wall. You press the tape so part of it sticks to the poster and the rest sticks to the wall. The tape enters any microscopic pits in the wall. Adhesive forces between the tape and the wall hold the poster in place.
Cohesive forces between the molecules within the tape’s adhesive layer are important as well. If the cohesive forces are too weak, the molecules within the adhesive layer would “let go” of each other, and the poster would fall to the floor.
How Tape Works
How Tape Works
Back to nature
Sticky chemistry is pretty well established. But there are still things most synthetic adhesives can’t do. For example, synthetic adhesives are not very good at making things stick together underwater. To advance sticky chemistry even further, scientists are again looking to nature for inspiration. Living organisms often secrete adhesives for attachment, construction, defense, and predation. They have the benefit of millions of years of evolutionary experience in developing their “products.” Species such as mussels and barnacles do this easily—even under harsh conditions.
Unlike early humans’ adhesives, however, new solutions probably won’t involve using animal and plant products but will mimic them. For example, reseachers at Purdue University created a biomimetic (copying nature) synthetic polymer after examining mussels’ protein-based adhesives. Mussels use tiny hair-like fibers covered in an adhesive to attach to surfaces. The mussel’s glue contains the amino acid dihydroxyphenylalanine (DOPA), which gives it a unique adhesive strength. DOPA contains a reactive functional group known as a catechol, which consists of two hydroxyl groups sticking out from a benzene ring. The catechol group interacts with surfaces, such as rocks, through intermolecular forces including hydrogen bonding. By combining the chemistry of mussel proteins and DOPA into a new polymer, the Purdue researchers created a strong underwater adhesive.
Slug slime and gecko feet have also inspired the development of sticky experimental products. Researchers say that one day these biomimetic adhesives could help surgeons fix organs or help astronauts walk on the outside of a spacecraft!
What amazing adhesive applications can you imagine?
Getting Rid of Unwanted Adhesives
Peeling price-tag stickers off newly purchased books, mugs, and binders often leaves a gummy residue behind. Scraping off the tacky adhesive with a fingernail doesn’t always do the trick, but there’s another way to remove the stickiness.
Remember the rule of thumb: “Like dissolves like”? That is, polar substances dissolve in polar media, and nonpolar substances dissolve in nonpolar media. The residue from price tags and labels is typically made of non-polar, polymeric molecules. So, nonpolar products will dissolve the nonpolar residue, which would allow you to remove it from your new purchase. For example, limonene, a naturally occurring compound in oranges, is a nonpolar organic molecule that is often used in cleaning products.
Unsticky Hack
Ingredients:
- 1 part baking soda (Sodium bicarbonate, NaHCO3, as an abrasive agent; it also helps make a non-runny paste)
- 1 part melted coconut oil (rich in nonpolar fatty acids of medium size; excellent for dissolving nonpolar goo)
- sweet orange oil (or other citrus essential oils)
Combine ingredients, adding just enough baking soda to the coconut and orange oils to make a paste. Apply the paste. Wait an hour or two, and peel or scrub off the adhesive!
REFERENCES
Kingsley, G. R. Masonry. Encyclopedia of Physical Science and Technology, 3rd Ed.; Meyers, R. A., Ed.; Academic Press: Cambridge, 2003; pp 133–143: https://doi.org/10.1016/B0-12-227410-5/00405-1 [accessed Aug 2020].
Kozowyk, P. R. B. et al. A New Experimental Methodology for Assessing Adhesive Properties Shows that Neandertals Used the Most Suitable Material Available. Journal of Human Evolution, Dec 2019: https://doi.org/10.1016/j.jhevol.2019.102664 [accessed Aug 2020].
Steven, E. Meet the Determined Woman Who Invented Duct Tape. Johnson & Johnson, Feb 8, 2018: https://www.jnj.com/our-heritage/vesta-stoudt-the-woman-who-invented-duct-tape [accessed Aug 2020].
Thanks to Elayne Thomas and Hares Wahdat, postdoctoral researchers at the University of Massachusetts, Amherst, for their assistance with this article.