Marshmallows seem simple when you look at them on their powdery, sugary surface. “A marshmallow is a yummy, chewy, airy confection that takes advantage of many unique food-science properties,” says Linda Wright, director of food research and discovery at the Hershey Company. “All the ingredients contribute to the delightful texture and flavor of the experience of a marshmallow.”
Whether you make them yourself or buy a bag of them, these puffy little sweets have magic to them because of the chemistry of the ingredients and how they interact.
The Key to a Gooey Marshmallow
A typical marshmallow contains sugar, corn syrup, and gelatin, plus some air. That’s it.
“A marshmallow is basically a foam that’s stabilized by gelatin,” says Richard Hartel, a food engineer at the University of Wisconsin–Madison.
In marshmallows, the foam is made up of air suspended in a liquid sugar mixture.
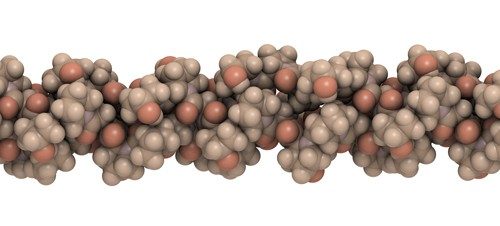
Gelatin is probably the most important part of a marshmallow, because it serves as the scaffolding that gives the marshmallow its stretchy, gooey texture. Gelatin is made by partially breaking down collagen, the main connective material in animal tissues, into smaller protein molecules.
“Collagen is a triple helix. It’s a helix of helices,” says Matt Hartings, a bioinorganic chemist at American University in Washington, D.C.
Collagen: A Helix of Helices
When you’re making marshmallows, one of the first things you do is add the gelatin to warm water. The heat breaks down the bonds holding the helices’ polypeptide chains together, causing the protein strands to partially unravel and spread out in the water, Hartings says. As the water starts to cool, parts of the gelatin molecules wind back together in threes, making up that helix of helices. Some parts of the gelatin, however, don’t wind back together and instead remain floppy and flexible. This structural mix of bendy and firm parts is what gives gelatin its elasticity, perfect for making bouncy foods like marshmallows.
This elasticity is hard to simulate, Hartel says. Vegan marshmallows do exist, and they use soy protein and carrageenan instead of gelatin. Carrageenan, which comes from seaweed, consists of chains of polysaccharides that form helices. Together with soy protein, it gives the ani-mal-product-free marshmallows a bouncy texture similar to that of gelatin-based marshmallows, although not quite as firm.
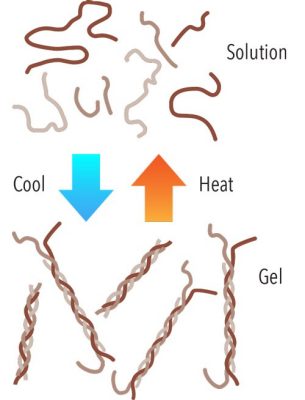
Another useful characteristic of gelatin is that it forms a thermo-reversible gel, which means that it can flip back and forth between liquid and gel depending on the temperature.
“The melting point of gelatin is about 95 °F (35 °C), which is just below normal body temperature,” Wright said in an email. “This contributes to how marshmallow melts smoothly in the mouth when eaten.”
Candymakers can change the texture of gelatin-containing foods by varying how much gelatin they use, Hartel says. For example, marshmallows and gummy bears are both made of gelatin, corn syrup, and sugar.
“We use more gelatin in a gummy bear than we do in a marshmallow, because we want the gummy bear to have firm characteristics,” he says. In a gummy bear, the gelatin forms a gel structure by itself. In a marshmallow, there’s another key component: air.
Fantastic Elastic
Gelatin comes from collagen that’s been partially broken down by either an acidic or an alkaline treatment and then purified. The elastic gelatin strands can then transition between a solution and a gel, unraveling under heat and twining back together when cooled.
Creating Fluff and Sweetness
To make a marshmallow, you whip air into the gel between its melting and solidifying stages so that the gel grabs the air and enmeshes it in a tangled 3D polymer, Hartel explains.
“If you’ve done it right, you whip [the mixture] just above the melting point of the gelatin. Then it cools below the gel point, and it solidifies,” Hartel says.
The solidified gelatin matrix then holds onto the air whipped into it to stay puffy and soft. You could whip air into a gummy-bear mixture and make it big and billowy too, Hartel says. How much air would it take to puff up this stiffer mixture? “A lot,” he says.
But of course, while vital to the marshmallow’s fluffiness, air is not the only thing trapped inside. It wouldn’t be a treat without the sweet stuff, specifically sugar and corn syrup.
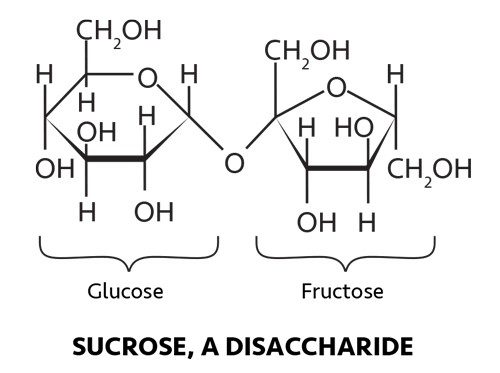
The two sweeteners are actually different. Sugar, or sucrose, is made up of one glucose and one fructose molecule linked together. When two simple sugars are joined to form one larger carbohydrate, the resulting molecule is called a disaccharide. Corn syrup contains a mixture of the sugars, such as maltose, glucose, and a variety of polysaccharides derived from corn starch.
Playing with Texture
Corn syrup pulls double duty in a marshmallow. It doesn’t crystallize like sugar does, plus it actually reduces the amount of dissolved sugar that crystallizes. A marshmallow with no sugar, only corn syrup, would be much less sweet because corn syrup is not as sweet as sugar, Hartel says. Corn syrups can also have varied amounts of different saccharides, so the type of corn syrup used also matters, Wright says. “It can provide a range of textures from chewy to tender depending on the type of corn syrup selected when mixed with sucrose.”
How much of each sweetener you add can make or break the texture of the marshmallow, because the syrup can keep the sugar from crys-tallizing. Sugar crystals tend to make confections more brittle than stretchy, a texture known as “short,” Hartel says. These grained marshmallows, such as Spangler Candy’s Circus Peanuts, tend to be harder and less fluffy.
“The difference in formulation between something like a Circus Peanut and a Jet Puffed [marshmallow] is the ratio of sucrose to corn syrup,” Hartel says. A Circus Peanut has more sugar and less corn syrup, while in marshmallows it’s the other way around.
Last but not Least: Peeps!
Some of the most famous varieties of marshmallows are Peeps, the sugar-covered treats shaped like chicks and bunnies made by Just Born. People tend to get creative with the brightly colored marshmallow candies, from crafting dresses and science-themed dioramas, to performing lab tests on them. There’s also a sport called Peeps jousting. The rules are simple. Two Peeps face each other in a microwave, and each has a toothpick pointing at their opponent. You then microwave the combatants for around 40 seconds (depending on the strength of your microwave). The Peeps inflate as they heat up. The first one to poke the other wins. You can do this with regular marshmallows, too, but the results aren’t as cute.
As you press the start button on the microwave, a lot of things happen at once, Wright says.
“Water molecules start to vibrate and heat and soften the sugar matrix. Also, the air bubbles heat up and expand. Since the sugar matrix surrounding the bubbles is softened, the bubbles are allowed to expand. The marshmallow expands and puffs up,” Wright said in an email. The Peeps’ behavior is due to the ideal gas law, Hartel says. The law describes the relationship between pressure, temperature, volume, and amount of gas.
But as the candy puffs up, some of the air bubbles burst, causing the marshmallow to deflate like a popped balloon, Wright says.
“When the marshmallow cools, the bubbles shrink, and the sugar matrix hardens,” which is why microwaved Peeps can get hard as rocks.
“Additionally, the loss of water can make the texture crunchy.”
Whether they’re jousting them or just eating them, people seem to like Peeps. They are the best-selling nonchocolate Easter candy, according to Just Born, and in 2021 the company teamed up with PepsiCo to make a marshmallow-flavored soda. Chugging a limited-edition “Peepsi,” however, wouldn’t have recreated the fluffy, gooey, exploding sweet puffball of a real marshmallow. And we have food chemistry to thank for these fluffy treats.

Refresher on the Ideal Gas Law
The ideal gas law is a summary of many observations on the relationship between pressure (P), temperature (T), volume (V), and number of moles of a gas (n). The relationship is often written as follows:
PV = nRT
R = gas constant
The ideal gas law helps us understand and predict gas behavior under most conditions. It assumes, however, that all gas molecules are the same size and experience no attractive and repulsive forces. It is less helpful for understanding gases at very low temperatures or very high pressures.
This article was adapted from “What’s in Marshmallows, and How Do the Ingredients Work Together to Make Ooey-Gooey Treats?”, which was originally published in Chemical & Engineering News on April 2, 2021.