Cheesy Science
By Sarah Mullen Gilbert December 2017/January 2018

Did you hear about the explosion at the
cheese factory?
There was de brie everywhere!
Ba dum bump. Thank you, thank you—we’ll be here all article. Now, get ready for some cheesy science and maybe a few more cheesy jokes.
We love cheese. Love it, love it, love it. And we are eating more of it—in the past 30 years, the average cheese consumed per person in the United States has increased 41%, up to 36 pounds of cheese per person. Mozzarella consumption was up 178% in the same period.
But cheese is kind of weird. It is storable milk. It can last weeks or years longer than milk and there is such a variety of cheese. But when did we start eating cheese? We don’t know the exact origin but at some point, someone must have thought, “You know that stuff at the bottom of the milk? That stuff that dried out and sat for weeks? I’m going to eat that!” And when they survived, proto-cheese was born.
Humans have used practical knowledge of chemistry and biology to preserve food for millennia, long before refrigeration and other modern technology. To make cheese, bacteria digest sugars in milk and produce lactic acid. The additional lactic acid lowers the pH and hinders the growth of harmful organisms. By turning milk into cheese, its shelf life is extended from about three weeks to two decades, or even longer.
All cheese starts as milk. The primary sources are milk from cows, goats, and sheep, though other mammals such as water buffalo are also used around the world for milk production.
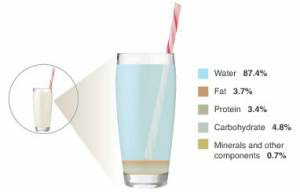
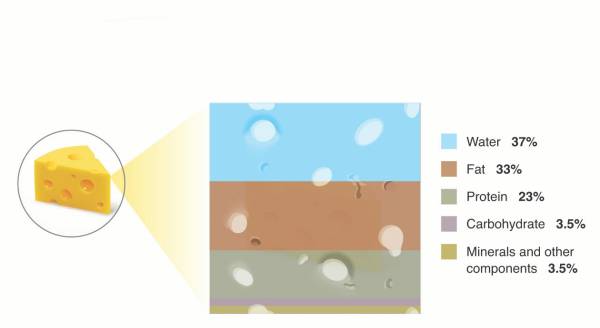
Figure 1. Cow’s milk compared to hard cheddar. Cow’s milk is mostly water; cheesemaking removes much of the water and concentrates the remaining fat, protein, and other components.
Basically, Basic Cheese is Acidic. What?!
The basic steps in cheesemaking are: adding beneficial bacteria to milk, coagulating the milk into a soft white substance called curd, and pressing and cutting curd into the finished cheese shape. But to get a delicious final product, the milk must be at the right temperature and the right pH at the right time.
“You cannot fully understand cheese and cheesemaking unless you understand acidity and pH,” says Paul Kindstedt, professor at the Vermont Institute for Artisan Cheese, in Burlington, Vt.
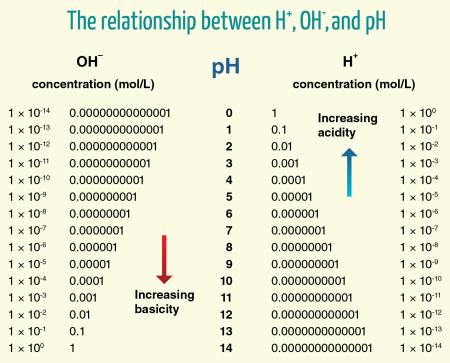
The pH measures the concentration of hydrogen ions (H+) in a solution, with most solutions between the range of 0 and 14. The more acidic the solution, the lower the pH, with 7 considered “neutral” and above 7, “basic.” Milk has a pH between 6.6 and 6.7. pH is a logarithmic scale, so one unit differs by a factor of 10. A pH of 6 has ten times the H+ concentration than a solution with a pH of 7. This logarithmic scale is why even apparently subtle pH differences can be quite significant.
To make cheese, milk is pumped into a large tank and warmed to the right temperature. Two different bacteria are used in this process: mesophilic and thermophilic bacteria. Mesophilic bacteria grow best in moderate temperatures, typically between 20 and 45°C (68 and 113 °F). They are used to make mellow cheeses such as cheddar, gouda, and Colby. Thermophilic bacteria thrive between 45 and 122 °C (113 and 252 °F) and are used to make sharper cheeses such as Gruyère, Parmesan, and romano.
In the tank, the bacteria ferment the sugar present in milk, called lactose (C12H22O11), into lactic acid (CH3CHOHCOOH), as follows:
C12H22O11 + H2O ⇾ 4 CH3CHOHCOOH
As more lactic acid is produced, the milk’s pH lowers. “pH is our indicator of activity,” says Jeremy Stephenson, cheesemaker at Spring Brook Farm in Reading, Vt. “When the pH changes, we know the bacteria are alive and well. By measuring the pH, we are measuring the activity of these bacteria and assuring that the fresh curd is on the right path to becoming cheese.”
After the bacteria replicate and culture the milk at the optimal temperature, the milk coagulates and changes from a liquid into a firm, rubbery material. This change takes an hour or two and is possible because of the casein proteins in milk. Casein molecules aggregate into spheres called micelles. The outer layer is negatively charged, which allows the micelles to remain dispersed in liquid milk. To form cheese, the proteins must coagulate, or stick together (Fig. 2).
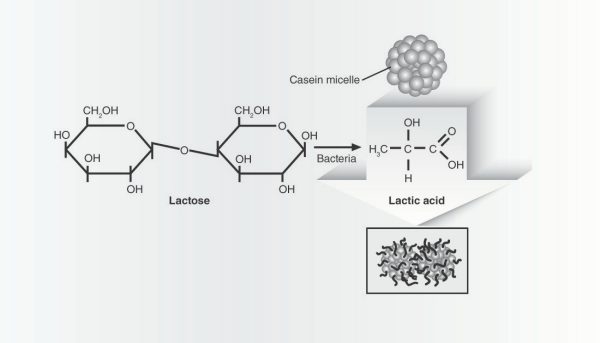
Figure 2. Casein comprises 80% of the protein in milk. The casein molecules are normally wrapped into compact spheres that are packed together with calcium and phosphate ions to form microscopic micelles. Acid causes the casein molecules to partially unfold and link with each other. The interconnected micelles form a mesh-like structure that causes the milk to gel into a semisolid.
Soft cheeses, such as cream cheese, coagulate slowly. As bacteria produce lactic acid, the other layer of the casein micelle becomes less and less polar. The micelles begin sticking together at around pH 5.3, with full coagulation after 24 hours, at pH 4.6.
Hard cheeses, such as Colby and Swiss, require a faster coagulating phase and a firmer resulting curd, so cheesemakers add a substance called rennet. The chymosin enzyme in rennet cuts the negatively charged ends on the micelles’ surfaces. No longer polar, the micelles are repelled by water and begin sticking together. The micelles form chains, which extend in all directions and interlock into a three-dimensional matrix to trap the milk-fat molecules. The more acidic the milk (the lower the pH), the faster this coagulation occurs and the firmer the curd.
Rennet is found in a calf’s stomach—its fourth stomach. How were rennet’s properties discovered? Enzymes weren’t known about then. It is thought that an earlier forbear noticed that milk would prematurely curdle in containers made from calf stomach. Added pieces of dried stomach or an extract had the same effect and a cheese innovation was made.
Bacteria have an optimum pH range that allows them to grow; a solution outside that range will prevent bacterial growth. Similarly, enzymes work best for a specific range of pH values. To create the ideal environment for rennet enzymes, Gouda and cheddar should be coagulated at pH 6.55, while optimal coagulation for mozzarella and Brie cheeses are at pH 6.45.
Once the milk has coagulated, the resulting curd is cut into smaller cubes, changing from a rubbery solid into cubes floating in liquid whey. Whey does not precipitate as the pH drops because of the proteins present. Whey proteins do not contain phosphorus, so they remain dissolved in the watery part of milk. As a result, liquid whey is expelled, removing water and further concentrating the remaining milk components. Whey is the liquid by-product of cheese making, contains soluble proteins, and is sometimes used as a substitute for skim milk or as a fertilizer.
As curds expel more whey, the result is a drier final cheese. For some varieties of cheese, such as Swiss cheese, curds are heated to further remove moisture.
Cheese curds are transferred into molds to drain completely and to give the cheese its final shape. Depending on the type of cheese, the curds are treated differently, giving it the shape and consistency of the cheese you buy at the store or at a farmer’s market. Soft, spreadable curds with low pH are ladled into bags to hang overnight and drain. Curds for harder cheeses are shaped into wheels or large blocks and are pressed or weighed down. The added pressure drains additional whey and forces the curds together into the final cheese shape.

Different acidification rates
While all milk starts at about the same pH, and most cheeses finish at a similar pH, the rate of acidification, or how fast the pH drops, is vital for creating different types of cheese. This is a natural process that results from the fermentation. For example, when pressing Gouda cheese curds into molds, they should be at a pH of about 6.5; in contrast, mozzarella curds should be at pH 5.25. But when finished, the pH of Gouda is more acidic than that of mozzarella. The different taste and texture are accomplished through different rates of acidification, along with adding different culture organisms.
To get the right pH at the right time, cheesemakers rely on measurements. “It’s a mix of art and science,” says Krista Dittman, owner and cheesemaker at Branched Oak Farm in Raymond, Neb. “You need to understand the science and have measurement tools, but you also need to develop an intuition about when and what to do.” Dittman uses paper test strips to gauge the pH of cheeses at critical points in their development. This information guides the timing of steps in the process.
“We take lots of pH measurements while making mozzarella, but we only write down three: 1) start, 2) when rennet is added, and 3) the end,” Dittman says. “For a typical soft, spreadable cheese, such as quark, I spot-check the whey now and again.”
Some cheeses go right to store shelves, such as cream cheese and cottage cheese. Softer cheeses, such as Brie and Camembert, are aged for about two months. Harder cheeses can age for decades. Cheeses age in a temperature- and humidity-controlled environment for varying lengths of time depending on the type. As cheese ripens, bacteria break down the proteins, which alters the flavor and texture. The proteins first break into medium-sized pieces (peptides) and then into smaller pieces (amino acids). In turn, these can be broken down into highly flavored molecules called amines. At each stage, more complex flavors are produced.
Each of these cheeses starts with milk, but different bacterial cultures and different rates of acidification make each variety unique. Using lactic acid production to preserve food is a millennia-old process that still results in delicious food today.
Selected references
Aiken, K. The Top 10 Most Popular Pizza Toppings (Infographic).
The Huffington Post, Nov 12, 2013: http://www.huffingtonpost.com/2013/11/12/popular-pizza toppings_n_4261085.html
[accessed Sept 2017].
Lactose Intolerance: Causes. Mayo Clinic, Feb 9, 2016: http://www.mayoclinic.org/diseases-conditions/lactosezintolerance/basics/causes/
con-20027906 [accessed Sept 2017].
Sarah Mullen Gilbert is a science writer who lives in Iowa Falls, Iowa. This is her first article in ChemMatters.
Also in this Issue...
Open for Discussion:
Are Vitamin Supplements Necessary?

How would you choose whether you need a daily dose of a multivitamin?

Lactose Intolerance
Many lactose-intolerant people can eat aged cheese without discomfort. In the cheesemaking process, the bacterial cultures ferment milk’s lactose to produce lactic acid. The longer a cheese ages, the less lactose is left over because it’s been used by the bacteria. By the time an aged cheddar or Parmesan is ready to eat, hardly any lactose remains in the cheese.