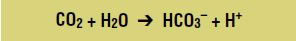24 Hours: Your Food on the Move
By Brian Rohrig
ChemMatters February 2012
- Download article
- Teacher’s Guide
WORD │ PDF
After you eat, you probably don’t give your food another thought. But the story of what happens to your food after you swallow it is quite fascinating. The story of digestion actually begins in the brain, not in the mouth or the stomach. The smell, sight, or even thought of food is enough to increase salivation and get your gastric juices pumping. If you have ever watched a dog eyeing a steak dinner, you can literally see the drool (saliva) dripping from his mouth. People are usually a little more discreet than that!
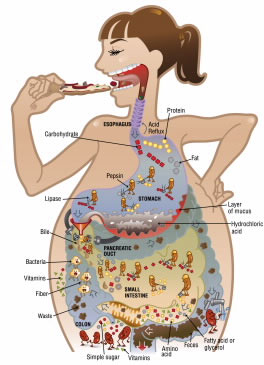
The digestive system looks like a narrow tube around 30 feet (9 meters) long, extending from the mouth to the anus. Digestion is the process of breaking food down into smaller and smaller pieces until it is in a form that can be used by the body. Think of it as a disassembly line that breaks materials down and discards parts that are of no use.
There are four main parts of the digestive process: ingestion, digestion, absorption, and elimination. Ingestion occurs when food is placed in the mouth. Digestion involves breaking down food, which takes place in the mouth, stomach, and intestines. Absorption primarily occurs in the small intestine, where anything of value is extracted and passed through the rest of the body. Then, whatever is left over is excreted as waste.
Ingestion
As soon as food enters your mouth, the digestive processes kick into action. Your teeth break food into smaller pieces—which helps to mix the food more thoroughly with the saliva—and you begin to chew these food pieces. Scientists have determined that the average mouthful of food is chewed 20 times before being swallowed. (Yes, there are scientists who study such things!) Some foods, such as carrots, may need to be chewed more, while others, such as nuts, fall apart rapidly upon chewing and need to be chewed less.
Saliva plays an important role in digestion. The three most important components of saliva are: water (98%), mucus, and digestive enzymes. Water moistens chewed food, and mucus lubricates it, making it slide down easily.
Saliva contains digestive enzymes that chemically break down food into a form the body can use. Enzymes are examples of catalysts—chemical compounds that speed up the rate of a chemical reaction without being consumed by the reaction. In other words, they can be used over and over again. There are thousands of different types of enzymes in the body.
To get an idea of just how fast some enzymes work, consider the enzyme carbonic anhydrase, which catalyzes a reaction that removes carbon dioxide from the cells before it goes to the lungs, where it is exhaled as waste. Each time a carbon dioxide molecule needs to be removed from the cell, carbonic anhydrase catalyzes the reaction that converts carbon dioxide and water molecules into a bicarbonate ion and a hydrogen ion:
Because bicarbonate ions are more soluble in blood than carbon dioxide, this reaction allows bicarbonate ions to be carried through the blood to the lungs, where the opposite reaction occurs, and the carbon dioxide is released by the lungs.
Carbonic anhydrase catalyzes the conversion of carbon dioxide into bicarbonate ions 600,000 times every second! It is no exaggeration to say that catalysts speed up the rate of a chemical reaction. Once the food is chewed, moistened, and lubricated, it travels down the throat, which leads into the esophagus and into the stomach. The tongue plays an important role in helping to transfer the saliva to the food and then rolling it into a ball, known as a bolus. The bolus doesn’t really roll down your throat but is helped by muscular contractions in the esophagus. You do not need gravity to swallow, or else the astronauts in space would be constantly hungry! You can actually swallow food while standing on your head, although this is not recommended.
Catalysts: Mountain-Climbing Made Easier
Catalysts speed up a chemical reaction by providing an alternate pathway for the reaction to occur. The role of a catalyst can be understood by the following analogy. Imagine that the reactants are climbing a mountain up one slope and then down the other slope to reach their destination. Adding a catalyst is like opening up a hidden pass in the mountain. The reactants won’t have to climb as high, so they don’t have to use as much energy, and they reach their destination faster.
Digestion kicks in once the food reaches the stomach. The stomach contains powerful muscles that churn food continuously, essentially beating the contents to a pulp. The stomach secretes juices that convert food into a liquid-like substance known as chyme. Most people know that the stomach contains acid, but fewer people know that this type of acid is hydrochloric acid. And fewer still know that the primary purpose of stomach acid is not to break food down but to provide an acidic environment that allows powerful digestive enzymes to do their work.
It is these enzymes, not the acid itself, that perform most of the chemical breakdown of food. Even if a piece of meat is placed in concentrated hydrochloric acid, it will not break down, but if you add enzymes, it breaks down quickly. Collectively, the combination of acid and enzymes is known as gastric juice, even though hydrochloric acid only comprises about 0.5% of the total volume of the gastric juice. Your body produces 2–3 liters of gastric juice daily.
The stomach, when completely full, can be stretched to hold about 2 liters of food. The average time for gastric emptying is about 4 hours; this time varies according to the quantity and type of food consumed. A light meal can be emptied from the stomach in about 2 hours, while a large heavy meal may require up to 6 hours. Forensic pathologists will often examine the stomach contents of a deceased person in an attempt to pinpoint the time of death.
To prevent the gastric juice from digesting the stomach itself, a thick layer of mucus coats the inside of the stomach. If the stomach produces too much acid, either due to eating too much or from a medical condition, heartburn can result. Heartburn has nothing to do with the heart, but rather is caused by gastric juice moving upward into the esophagus, creating a burning sensation.
This can be alleviated by taking antacids, which contain substances that neutralize acids. A common antacid is Tums, which contains calcium carbonate (CaCO3). Calcium carbonate neutralizes excess acid according to the following equation:

More Digestion, Absorption, and Elimination
Once the stomach has digested food, the liquid-like chyme passes into the small intestines. Additional enzymes produced by intestinal cells and by cells in the pancreas break down the leftover food further. Bile, secreted into the small intestine from the gall bladder, breaks up fat into tiny globules.
Most of the nutrients in food are absorbed by the small intestine and carried to other parts of the body. The walls of the intestine contain tiny blood vessels that transport nutrients first to the liver, where any toxins are removed before the nutrients are distributed throughout the rest of the body. On average, food spends 4–8 hours in the small intestine. Once the food leaves the small intestine pretty much anything of use has been extracted, and most of what remains is waste. This waste enters the large intestine, or colon. At this point, the waste is still very watery. A lot of this water will be absorbed through the walls of the colon. If the waste passes too quickly through the colon—for instance, if the colon is irritated by a virus—there is not enough time for the water to be absorbed, causing diarrhea.
A lot of fiber also causes food waste to pass through quickly. By the time the waste reaches the rectum—the last part of the colon—it is ready to exit the body through the anus, and it should have a somewhat solid consistency. Up to half of the weight of feces can be bacteria—living or dead—and their waste products.
Waste spends about 12 hours in the large intestine before it is excreted. Although most nutrient absorption occurs in the small intestine, the many bacteria within the colon feed on the fiber and produce additional nutrients that the body uses, such as vitamin K and vitamin B. Your body contains 3–5 pounds of bacteria, enough to fill a few soup cans! Most of the bacteria are found in the colon.
What is amazing about the digestive process is that once you have swallowed your food, the rest of the process is totally involuntary. Your body takes whatever morsel of food you give it and wrings out as many nutrients as it can. The digestive system is elegantly simple yet mind boggling in its speed, efficiency, and complexity. It is probably the best example of chemistry in action that you will ever experience, and it happens 24/7!
Selected references
- Snyder, C. H. The Extraordinary Chemistry of Ordinary Things, 4th ed.; John Wiley and Sons: Hoboken, NJ, 2003.
- Waldron, K. The Chemistry of Everything. Pearson Prentice Hall: Upper Saddle River, NJ, 2007.
- Wolke, R. L. What Einstein Told His Cook. W. M.Norton and Company: New York, 2002.
Brian Rohrig teaches chemistry at Jonathan Alder High School in Plain City (near Columbus), Ohio. His most recent ChemMatters article, “Titanic: Was It Doomed by Chemistry?” appeared in the December 2011 issue.
Also in this issue (February 2012)

A Tan – Quick, Easy, and Safe?
What is the best way to get a tan? We look at the various options—sun tan, tanning beds, and tanning creams and lotions—and discuss their advantages and potential safety issues.

Attack of the Gluten
Some people have skin rashes and stomach cramps when they eat foods that contain gluten, a substance found in wheat, barley, and rye. Why are these people sensitive to gluten, and what can they eat instead?