By: Louis J. Diorazio, Chemical Development, Pharmaceutical Technology & Development, Operations, AstraZeneca, Macclesfield, United Kingdom

In the heat of scientific progress and corporate challenges, the role of solvents is often downplayed. It can be easy to assume that a solvent merely dissolves materials, indeed, awareness of solvents for the general public is generally restricted to situations such as cleaning a stain or grease with ‘a bit of solvent’.
Although this is one use, solvents have a massive role on the success and impact of their application. Amongst other things, a solvent will dictate the acidity of a mixture and whether a chemical transformation might be supported or inhibited. The physical properties of a solvent are defined by the chemical structure. This is most apparent for water, which is famously atypical in its behaviour. The extensive hydrogen-bonding available due to its structure supports abnormally high melting and boiling points, enthalpy of vaporisation, surface tension and critical point relative to similar-sized molecules. And fortunately so, since this unique behaviour is critical to the existence of life on earth.
Choosing a solvent should always start with the question ‘Does a solvent benefit my process?’ Basic economics dictates that solvent use in ultra-high volume manufacturing should be minimal. For more specialty applications such as pharmaceuticals, solvent use can be significant. Analysis within the ACS Green Chemistry Institute Pharmaceutical Roundtable (ACS GCIPR) and elsewhere shows that solvents and water comprise over 70% of materials processed in pharmaceutical processing.1 Since the manufacture of 1 kg of active pharmaceutical ingredient (API) might consume 100s of litres of solvents, the environmental impact of such material consumption must be accounted for. As sustainability goals have been prioritized in companies, chemists and engineers have had to recognise that their choices in selecting materials to apply within chemical processes shape the environmental footprint of their companies.
About five years ago, some colleagues and I at AstraZeneca published our philosophy on solvent selection and details of a tool based on the data visualisation application, Spotfire2. Once a need for a solvent was established, the tool supported an approach that asked ‘What properties do I want from my solvent?’ and only then ‘What solvents meet my requirements?’ Unlike previously published tools intended for post-selection corroboration, ours incorporated a green assessment during the initial solvent selection process.
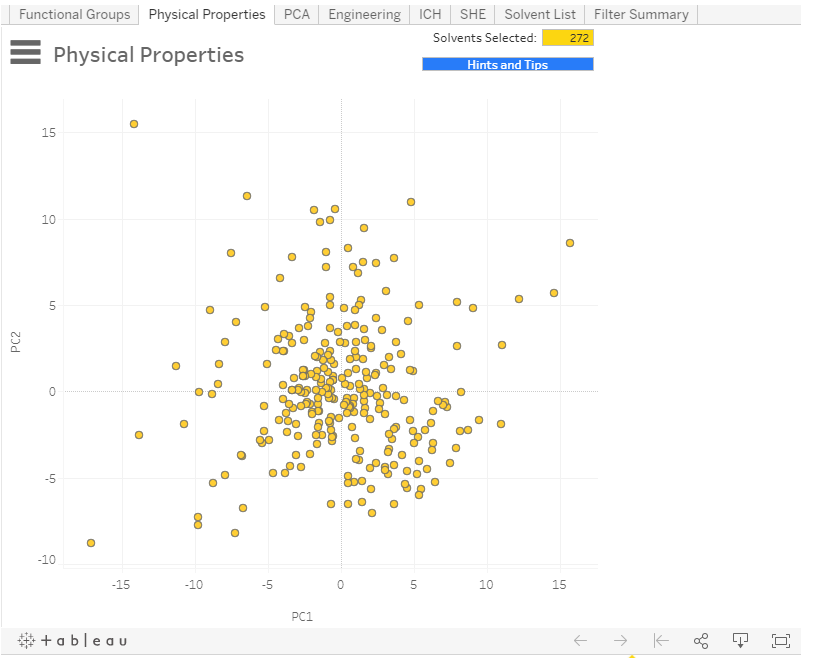
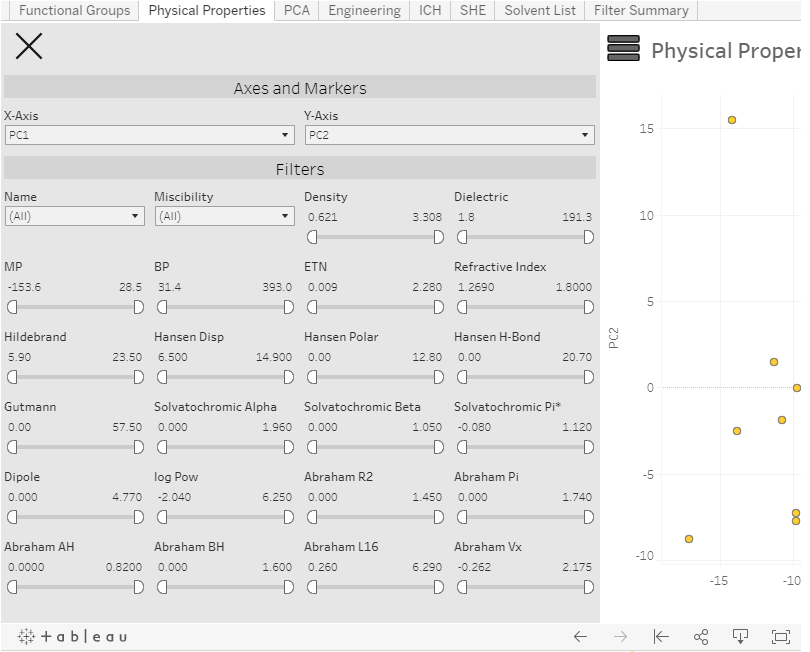
Over 270 potential solvents could be interrogated based on application needs such as chemical functionality and physical properties alongside a green assessment and the more pharmaceutically-focused concern of International Council for Harmonisation (ICH) classification. Several undesirable solvents such as carbon tetrachloride and HMPA were included since we recognised the need to identify alternatives to such materials, e.g. when encountered in the older chemical literature.
A visual distribution based on principal component analysis identified whether a solvent of interest had close-lying alternatives with similar properties or was more distinct from other solvents. In order to reduce bias and support identification of less familiar but otherwise acceptable solvents, the solvent display was anonymised until the final shortlist reveal, although identifying rollover labels were included. The dynamic nature of the tool supported rational contraction or expansion of the shortlist, supporting interrogation through lab studies or further refinement using in silico methods.
The response to the publication and associated presentations identified significant 3rd party interest in access to the tool. AstraZeneca is a longstanding member of ACS GCIPR and we donated the tool for further development with the objective of an open access version. The ACS GCIPR Solvent Selection Tool was delivered as a web tool in 2018 based closely on the original AstraZeneca version and containing 272 solvents. It is available from the ACS GCIPR green chemistry tools website alongside the Reagent Guide and other open access ACS GCIPR tools.
In the meantime, ACS GCIPR had also taken on stewardship of the legacy tools from the EU-funded CHEM 21 consortium. This included an algorithm-based approach to assessing the green credentials of a solvent based on a combination of readily available physical properties and GHS risk phrases. In 2019, the Solvent Selection Tool was upgraded to include this assessment such that complementary tools offered by ACS GCIPR were harmonised in their approach.
Since its launch, the Solvent Selection Tool has received almost 50,000 hits with good feedback from both industry and academia. It offers a structured framework that can support a response to legislative controls such as REACH whether demonstrating a rigorous selection process (and 272 solvents suggests casting a wide net) or seeking to identify replacements.
There is no instant panacea; introduction of a greener solvent to a poor process does not instantly mitigate other weaknesses that compromise sustainable performance. Solvent selection is an essential component of designing greener processes; the days of simply defaulting to whatever is in the lab store cupboard or the traditional choice should be long gone. Sustainable process development requires the choice of all input materials to be comprehensive - cost, performance, environmental, legal, etc. The Solvent Selection Tool demonstrates a solution to such problems.
- Hargreaves, C. R. M., J. B. ACS GCI Pharmaceutical Roundtable: Collaboration to Deliver a Solvent Selection Guide for the Pharmaceutical Industry. http://www.acs.org/content/dam/acsorg/greenchemistry/industriainnovation/roundtable/solvent-selection-guide.pdf (accessed 26/03/2021).
- Diorazio, L. J.; Hose, D. R. J.; Adlington, N. K., Toward a More Holistic Framework for Solvent Selection. Organic Process Research & Development 2016, 20 (4), 760-773.
This article has been edited for length and clarity. The opinions expressed in this article are the author's own and do not necessarily reflect the view of their employer or the American Chemical Society.
Copyright 2022 American Chemical Society (All Rights Reserved)








