What molecule am I?
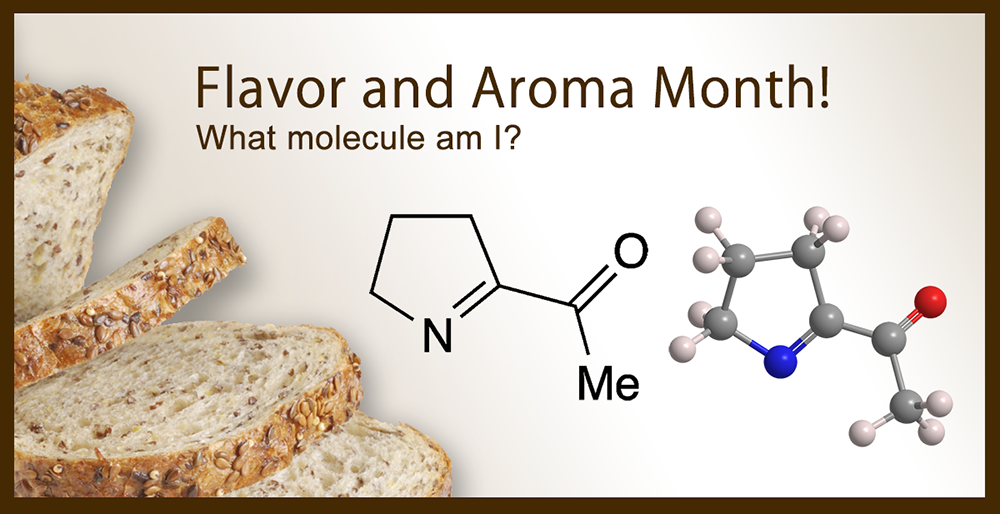
November is flavor and aroma month at MOTW!
2-Acetyl-1-pyrroline (2AP) is a ketone derivative of the heterocyclic compound 1-pyrroline. The hazard information table shows that the pure compound has some dangerous properties, but you probably know it from its pleasing aroma.
In 1982, Ron G. Buttery and Louisa C. Ling at the US Department of Agriculture (Albany, CA) and Bienvenido Juliano at the International Rice Research Institute (Los Baños, Philippines) identified 2AP as an important aroma component of cooked rice. It is present in the volatile oils of basmati and Calrose rice. The researchers also synthesized 2AP via the catalytic reduction of 2-acetylpyrrole, followed by oxidation with silver carbonate.
The following year, the same authors identified 2AP in the leaves of the tropical plant pandan (Pandanus amaryllifolius). The leaves are used as a flavoring agent in south and southeast Asia.
Later in the 1980s, chemists began to identify 2AP in the aroma of baking bread. In 1985, Peter Schieberle and Werner Grosch at what is now the Technical University of Munich (Garching, West Germany) reported that the compound, in addition to other flavor and aroma ingredients, is present in the crusts of wheat and rye breads. 2AP is formed by the Maillard reaction that is characteristic of baking bread, in which amino acids and reducing sugars react to form the components of the crust’s brown color.
Curiously, on a less appealing note, 2AP also contributes to the urine odor of several Asian mammals, including the bintarong (Arctictis binturong; aka the bearcat) and three subspecies of tiger (Panthera tigris).
2-Acetyl-1-pyrroline hazard information
| Hazard class* | GHS code and hazard statement | |
|---|---|---|
| Flammable liquids, category 2 | H225—Highly flammable liquid and vapor | |
| Aspiration toxicity, category 1 | H304—May be fatal if swallowed and enters airways | |
| Skin corrosion/irritation, category 2 | H315—Causes skin irritation | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
| Reproductive toxicity, category 2 | H361—Suspected of damaging fertility or the unborn child | |
| Specific target organ toxicity, repeated exposure, category 2 | H373—May cause damage to organs through prolonged or repeated exposure | |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals. Explanation of pictograms.
Molecule of the future
Polybutylene adipate-co-terephthalate1 (PBAT) is a copolymer that consists of two units: the 1,4-butanediol monoester of terephthalic acid and the 1,4-butanediol monoester of adipic acid (hexanedioic acid). The two units are dispersed randomly throughout the length of the copolymer.

PBAT has gained much traction recently because it is a potential biodegradable replacement for conventional plastic polymers. Even though all of its components are petrochemicals, the copolymer’s ester linkages give it decomposition properties similar to those of two other developmental biodegradable polymers, poly(lactic acid) and poly(hydroxyalkanoate).
The physical properties of PBAT are comparable with those of low-density polyethylene, which is used to make stretchable films for food-packaging bags, trash bags, and the like. Corporations worldwide are moving rapidly to increase production of PBAT.
1. CAS Reg. No. 60961-73-1.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
2-Acetyl-1-pyrroline
fast facts
| CAS Reg. No. | 85213-22-5 |
| SciFinder nomenclature | Ethanone, 1-(3,4-dihydro-2H-pyrrol-5-yl)- |
| Empirical formula | C6H9NO |
| Molar mass | 111.14 g/mol |
| Appearance | Colorless to yellow solid or liquid |
| Melting point | 19° C |
| Boiling point | 183° C |
| Water solubility | Soluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

