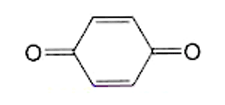
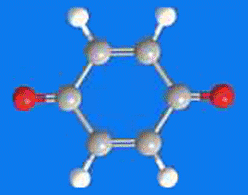
1,4-Benzoquinone (or, less formally, “quinone”), is a yellow crystalline solid with a chlorine-like odor. It is the simplest member of the quinone family of conjugated dienediones that have several uses in organic chemistry. It undergoes typical ketone and olefin reactions, but its predominant use is as an oxidizing agent. 1,4-Benzoquinone and its reduced form 1,4-dihydroquinone (1,4-dihydroxybenzene) form the basis of many redox systems.
MOTW update:
January 31, 2022
1,4-Benzoquinone1 (aka quinone) is a useful oxidizing agent and, along with its reduced form, hydroquinone2 (1,4-benzenediol), is the basis of many redox systems.
This month, Qinglei Meng, Buxing Han, and collaborators at the Chinese Academy of Sciences (Beijing) and East China Normal University (Shanghai) reported an innovative electrochemical method for simultaneously producing benzoquinone and cyclohexanone3 from phenol and water. In the authors’ electrochemical system, the cathode consists of nickel and platinum supported on nitrogen-doped hierarchically porous carbon (NHPC), whereas the anode contains iron and ruthenium, also supported on NHPC. Benzophenone and cyclohexanone form at the cathode and anode, respectively, both with >99.9% selectivity.
1. CAS Reg. No. 106-51-4.
2. CAS Reg. No. 123-31-9.
3. CAS Reg. No. 108-94-1.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

