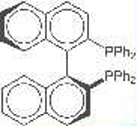
The two BINAP enantiomers [(S)-BINAP is shown] are widely used as ligands in enantioselective organic synthesis. BINAP has no specific stereogenic centers; its chirality stems from sterically restricted rotation about the bond that connects the naphthalene rings. The 2001 Nobel Prize in Chemistry was awarded to Ryoji Noyori in part for his group’s development of this technology.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

