What molecule am I?
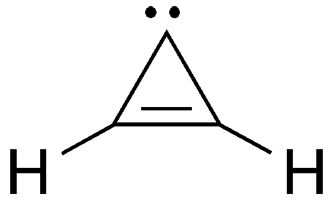
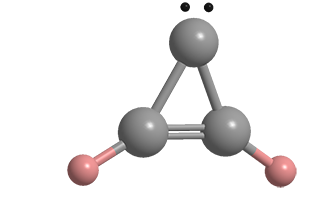
Cyclopropenylidene is a simple carbene, a molecule with a carbon atom bearing a pair of unshared electrons. Almost all carbenes, including this one, are unstable; but their presence is verified by the structures of the products they form when they react with other compounds.
The existence of cyclopropenylidene was first reported in 1965, when William Maurice Jones at the University of Florida (Gainesville), in a monograph, described its synthesis and reactions. He classified the molecule as a “non-benzenoid aromatic”. Jones and colleagues went on to study substituted cyclopropenylidenes.
Although they cannot be isolated, cyclopropenylidene and many other carbenes have been proven useful in organic synthesis. But it took their discovery in outer space to make it possible to observe them as free molecules. In 1985, Patrick Thaddeus*, Jan M. Vrtilek, and Carl A. Gottlieb at the NASA Goddard Institute for Space Studies at Columbia University (New York) found cyclopropenylidene in two locations: in the star Ori A in the belt of the constellation Orion and in the molecular cloud Sgr B2 in the constellation Sagittarius1.
Closer to home, in 2020 Conor A. Nixon at NASA Goddard Space Flight Center (Greenbelt, MD) and nine coauthors worldwide reported the presence of cyclopropenylidene in the atmosphere of Titan, Saturn’s giant moon. It was the first time the molecule was found in any atmosphere, Solar System or elsewhere. The researchers used the Atacama Large Millimeter/submillimeter Array (ALMA) radiotelescope in Chile to identify cyclopropenylidene in Titan’s upper atmosphere, where there are relatively few gases.
The spectral observations at ALMA were made in 2016 and 2017, but the authors took time to model the spectral emissions to verify that they were produced by cyclopropenylidene. The mean value of the molecule’s abundance from the two observations was 0.33 ± 0.07 ppb.
Because cyclopropenylidene is unstable on Earth, no physical property or hazard information is available.
1. Sgr B2 is one of the largest star-forming clouds in the Milky Way.
Molecule in the News
L-Tryptophan1 is an essential amino acid and the only one with a bicyclic structure in its side chain. It was the Molecule of the Week for November 21, 2017.
Tryptophan accounts for only 1% of the amino acids in proteins, but its shape is so different from that of other amino acids that researchers want to know how it interacts with amino acids in protein–protein combinations. To this end, F. Dean Toste, Christopher J. Chang, and co-workers at the University of California, Berkeley, sought a way to “tag” tryptophan to make it easier to examine its behavior.
Last month, the chemists reported an N-sulfonyloxaziridine molecule that selectively reacts with tryptophan and makes it easy to detect. Applying the reagent mimics oxidative cyclization reactions in indole-based alkaloid biosynthetic pathways. The new molecule also contains an ethynyl group that can be used to attach chemical or biological probes via “click” reactions.
1. CAS Reg. No. 73-22-3.
Molecule in the News
MOTW highlights molecules that appear in major news outlets. See this week's edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Cyclopropenylidene fast facts
| CAS Reg. No. | 16165-40-5 |
| CA Index Name | 2-Cyclopropen-1-ylidene |
| Empirical formula | C3H2 |
| Molar mass | 38.05 g/mol |
| Appearance | Gas |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

