What molecule am I?
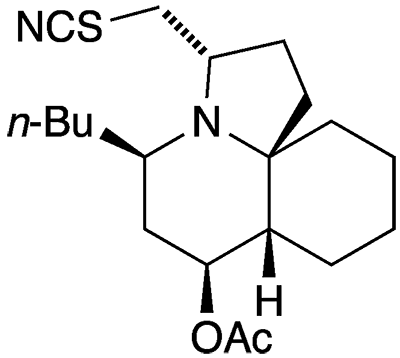
The cylindricines are a group of 11 marine alkaloids that were isolated off the coast of Tasmania from the ascidian1 Clavelina cylindrica between 1993 and 1995 by Adrian J. Blackman and co-workers at the University of Tasmania (Hobart). All the cylindricines have similar tricyclic pyrrolo- or pyrido[1,2-j]quinoline structures with varying functional groups (C=C, C=O, -Cl, -OH, -OMe, -OAc, -SCN, and/or -NCS) and alkyl side chains (n-butyl or n-hexyl).
Not all the cylindricines have fully worked-out stereochemistry or have been totally synthesized in the lab. But this past July, Mercedes Amat and co-workers at the University of Barcelona reported the total synthesis of (–)-cylindricine H2, which has both acetate and thiocyanate functionalities.
According to the authors, the key stereoselective steps were the generation of a quaternary stereocenter at C10; the introduction of the acetoxy group at C4 and the butyl group at C2; and the assembly of the pyrrolidine ring with the thiocyanatomethyl group in the proper configuration.
Because cylindricine H is not a commercial product, no hazard information has been reported.
1. Also known as a tunicate, an invertebrate sea animal.
2. Editor’s note: SciFinder gives the structure and nomenclature for cylindricine H as an isothiocyanate under the name 1H-pyrrolo[2,1-j]quinolin-7-ol, 5-butyldecahydro-3-(isothiocyanatomethyl)-, acetate (ester), (3α,5β,7β,7aβ,11aR*)-. This contrasts with the information reported in Blackman’s and Amat’s articles. In a personal communication, author Amat confirmed that thiocyanate is correct.
Molecules from the journals
1-Octen-3-one1 is an unsaturated straight-chain ketone with an odor that is both metallic and suggestive of mushrooms. This past September, John P. Munafo Jr. and fellow food scientists at the University of Tennessee (Knoxville) reported that 1-octen-3-one is by far the chief odor constituent of Cumberland rosemary (Conradina verticillata), an endangered shrub that grows only in Tennessee and Kentucky. Lesser odorants in C. verticillata include, among others, former Molecules of the Week eucalyptol2 (aka 1,8-cineole), bornyl acetate3, and eugenol4.
17α-Ethinylestradiol5 is a venerable estrogen derivative used in many birth control pills and other gynecological medications. It was first described in 1938 by Hans H. Inhoffen and Willy Logemann at Schering AG (Berlin) and approved by the US Food and Drug Administration in 1943.
17α-Ethinylestradiol, however, has an environmental downside. Last month, Jessica K. Leet at the Environmental Research Center of the US Geological Survey (Columbia, MO) and collaborators there and in Washington State and West Virginia reported the effects of the hormone on the economically important largemouth bass (Micropterus salmoides). When the juvenile fish were exposed to a low aqueous concentration (0.87 ng/L) of the compound, they were susceptible to bacterial infection. But unexpectedly, at a concentration of 9 ng/L, the fish had significantly increased survivorship.
1. CAS Reg. No. 4312-99-6.
2. CAS Reg. No. 470-82-6.
3. CAS Reg. No. 20347-65-3.
4. CAS Reg. No. 97-53-0.
5. CAS Reg. No. 57-63-6.
Molecules from the Journals
MOTW briefly describes noteworthy molecules that appeared in recent ACS journal articles. See this week's
edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Cylindricine H fast facts
| CAS Reg. No. | 164740-25-4 |
| Empirical formula | C20H32N2O2S |
| Molar mass | 364.55 g/mol |
| Appearance | Colorless oil |
| Melting point | Not determined |
| Water solubility | Not determined, but most likely insoluble |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

