What molecule am I?

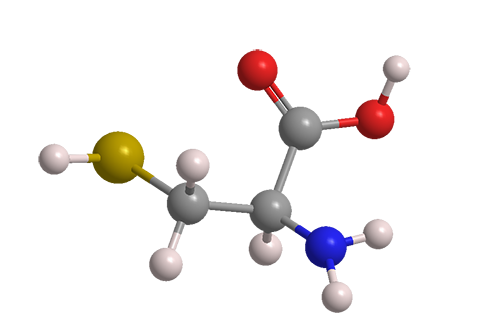
L-Cysteine is one of the two proteinogenic amino acids that contain a sulfur atom (the other is methionine). It is considered a nonessential amino acid because it is produced in the human body. Individuals who are deficient in cysteine, however, must be treated with supplements.
When cysteine is oxidized, the two thiol hydrogen atoms are eliminated, and the sulfur atoms join to form the dimer L-cystine. In biological systems, the monomer and dimer interchange easily; thus, both acids are nutritionally similar.
In September 2018, David Corey at Harvard Medical School and Jeffrey Hold of Boston Children’s Hospital reported that cysteine could play a role in improving hearing in humans. They discovered that the TMC1 protein in the inner ear’s hair cells forms pores that are involved in converting sound to electrical signals to the brain. In some individuals, a mutation in TMC1 can lead to deafness.
The researchers substituted residues in TMC1 with cysteine and then modified the cysteine groups with bulky charged groups. This artificial mutation caused the protein to cease the production of electric current, thus verifying that mutated TMC1 inhibits hearing.
These findings suggest that drugs that target TMC1 mutations or gene therapies that replace the mutated protein could help restore hearing to affected individuals.
L-Cysteine hazard information
| GHS classification*: not a hazardous substance or mixture |
*Globally Harmonized System of Classification and Labeling of Chemicals.
L-Cysteine fast facts
| CAS Reg. No. | 52-90-4 |
| Empirical formula | C3H7NO2S |
| Molar mass | 121.16 |
| Appearance | White crystals or powder |
| Melting point | 240 ºC (dec.) |
| Water solubility | ≈280 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

