What molecules are we?

Boranes are boron–hydrogen compounds, most of which contain delocalized electron bonds. Boranes with four or more boron atoms typically have completely or partially closed polyhedral cages. Examples are former Molecules of the Week tetraborane, pentaborane, decaborane, and cesium dodecaborate.
Similarly, carboranes are complete (closed) or partial borane polyhedra in which one or more of the boron atoms are replaced by carbon atoms. Carboranes have a nomenclature of their own: [n1,n2,n3, . . .][structural prefix][carbon prefix]carba[total atom prefix]borane, in which
- n1,n2,n3, . . . represents the position[s] of the carbon atom[s] in the polyhedron;
- the structural prefix indicates whether and how many vertices are missing from the polyhedron: closo- for complete, nido- for one missing vertex, arachno- for two missing vertices, and hypho- for three missing vertices;
- the carbon prefix shows the number of carbon atoms in the cage; and
- the total atom prefix represents the total number of boron and carbon vertices.
As an example, 2,3-nido-dicarbahexaborane is one atom short of a polyhedron with two carbon atoms in the 2- and 3-positions of a six-atom structure.
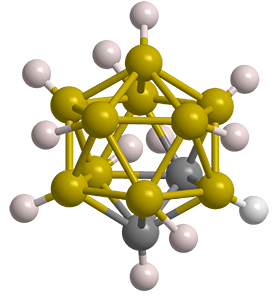
Of particular interest are the closo-dicarbadodecaboranes, which have two carbon atoms in complete 12-atom icosahedral structures. Of the three possible isomers, 1,2-dicarbadodecaborane is shown here. The other isomers have carbon atoms in the 1,7- and 1,12-positions. In some publications, the 1,2-, 1,7-, and 1,12-prefixes are denoted as o- (ortho-), m- (meta-), and p- (para-), respectively.
Unlike many boranes and carboranes, the closo-dicarbadodecaboranes are extremely stable. They are crystalline solids with high melting points and low solubility in water and organic solvents. They are somewhat interchangeable in that heating the 1,2-isomer to 420° C rearranges it to the 1,7-species, which upon additional heating to >600 °C rearranges to the 1,12-isomer.
1,2-Dicarbadodecaborane is prepared by the reaction of open-cage decaborane with acetylene, usually by first “priming” the decaborane with an adduct such as diethyl sulfide before the reaction with acetylene. 1,2-Dicarbadodecaborane derivatives are similarly synthesized by treating decaborane with substituted acetylenes.
Chemically, closo-dicarbadodecaboranes react similarly to organic cyclic compounds in that hydrogen atoms on carbon or boron vertices can be replaced by a wide range of substituents. For example, in a 1995 study, Peggy A. Radel and Stephen B. Kahl* at the University of California, San Francisco, used a several-step process to synthesize L- and D-enantiomers of 1,2-dicarbadodecaborane-substituted alanines. The alanine methyl groups were bonded to one of the carbon atoms on the dicarbadodecaborane cage.
Earlier this year, Alexander M. Genaev and co-workers at the N. N. Vorozhtsov Institute of Organic Chemistry (Novosibirsk, Russia) reported the noncatalyzed bromination of 1,2-dicarbadodecaborane at the 8- and 9-boron atoms on the icosahedral structure.
1,2-Dicarbadodecaborane hazard information*
| Hazard class** | GHS code and hazard statement | |
|---|---|---|
| Flammable solids, category 1 | H228—Flammable solid | |
| Acute toxicity, oral, category 4 | H302—Harmful if swallowed | |
| Acute toxicity, dermal, category 4 | H312—Harmful in contact with skin | |
| Acute toxicity, inhalation, category 4 | H332—Harmful if inhaled | |
*Compilation of multiple safety data sheets.
**Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
1,2-Dicarbadodecaborane
fast facts
| CAS Reg. No. | 16872-09-6 |
| SciFinder nomenclature | 1,2-Dicarbadodecaborane (12) |
| Empirical formula | C2H12B10 |
| Molar mass | 144.23 g/mol |
| Appearance | White crystals or powder |
| Melting point | 320 °Ca |
| Water solubility | Insoluble |
a. Melting points as low as 260 °C are reported.
MOTW/MOTF update
EIDD-2801 was the Molecule of the Week for June 1, 2020, and PF-07321332 was the Molecule of the Future for May 17, 2021. They showed great promise against SARS-CoV-2 infections. Both antivirals in pill form are currently in Phase 3 trials.
EIDD-2801, now named molnupiravir, is being evaluated by a partnership of Merck (Kenilworth, NJ) and Ridgeback Biotherapeutics (Miami, FL) as a treatment for patients with early COVID-19 and uninfected people who live with someone who tested positive for the virus. Pfizer’s PF-07321332 is also being tested on people who are unlikely to be hospitalized with the disease.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

