What molecules are we?
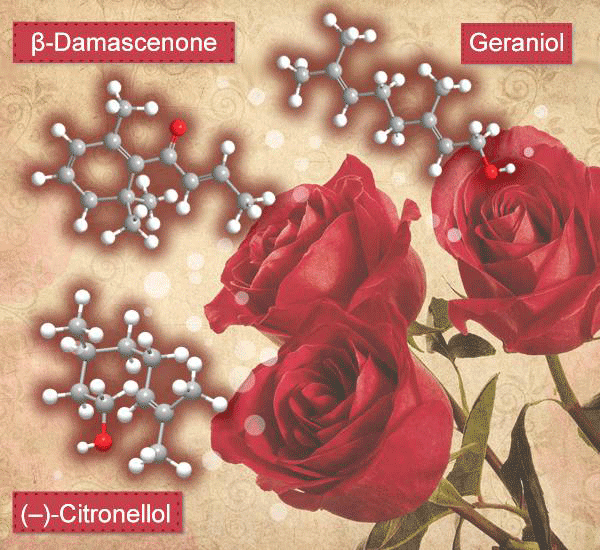
What is more evocative of Valentine’s Day than roses? But did you ever wonder about the compounds in roses that are responsible for the flower’s wonderful scent? Writing in the Journal of Chemical Education in 2011, Albrecht Mannschreck and Erwin von Angerer* at the University of Regensburg (Germany) gave a full accounting of roses’ aroma constituents.
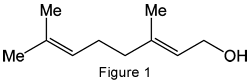
According to the authors, geraniol, (–)-citronellol, and β-damascenone (Figures 1, 2, and 3, respectively) are three major components of rose scent. Geraniol is monoterpene alcohol that is a main ingredient of rose oil; it also is present in many other essential oils. It was first isolated by O. Jacobsen in 1871. Its structure is similar to that of nerol, also a rose scent ingredient.
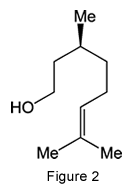
Citronellol exists in nature as two enantiomers, designated (+) and (–). The (+)-isomer is more common and takes its name from citronella oils. The (–) isomer, however, is the one that occurs in the oils of roses and geraniums. The citronellols are also monoterpene alcohols; they are the hydrogenated forms of geraniol.
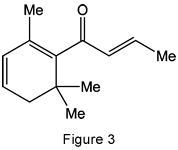
β-Damascenone is different: It belongs to a family of unsaturated ketones known as rose ketones. Its concentration in rose oil is very small, but it contributes significantly to the aroma.
All three of these compounds are articles of commerce as fragrance ingredients used by the perfume industry. So if you give your loved one perfume instead of roses for Valentine’s Day, she or he will likely get a whiff of one of them.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.