What molecule am I?

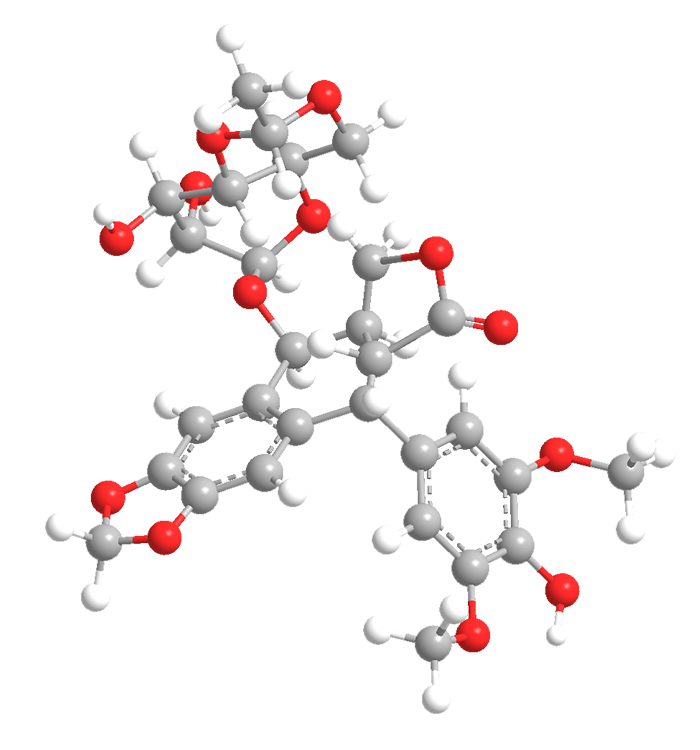
Etoposide is, by today’s standards, an ancient anticancer drug. In 1971, C. Keller-Juslén and co-workers at Sandoz reported its synthesis from the natural product podophyllotoxin, a toxic lignin found in the roots and rhizomes of plants of the Podophyllum genus. The US Food and Drug Administration approved it for use in 1983.
Etoposide is still being used and is still being made from podophyllotoxin. But the main source, the Himalayan mayapple (Podophyllum hexandrum), is difficult to grow; so Elizabeth S. Sattely and co-workers at Stanford University sought alternative sources.
The researchers found that they could transfer enzyme-producing genes from P. hexandrum to Nicotiana benthamiana, a wild tobacco-like plant that is easy to grow. So far, the engineered plants have produced only nanograms of etoposide aglycone, an etoposide precursor. Sattely believes that the same gene pathway can be introduced in to yeast, which reproduces prolifically and may provide greatly increased yields of the aglycone.
MOTW Update
The October 5, 2015, MOTW noted that a federal appeals court had overturned the registration of the insecticide sulfoxaflor because of its toxicity to honeybees. As a result of this court order, on November 12, 2015, the US Environmental Protection Agency stopped the sale of this chemical in the United States. The manufacturer, Dow AgroSciences, intends to work with EPA to return sulfoxaflor to the market.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

