What molecule am I?

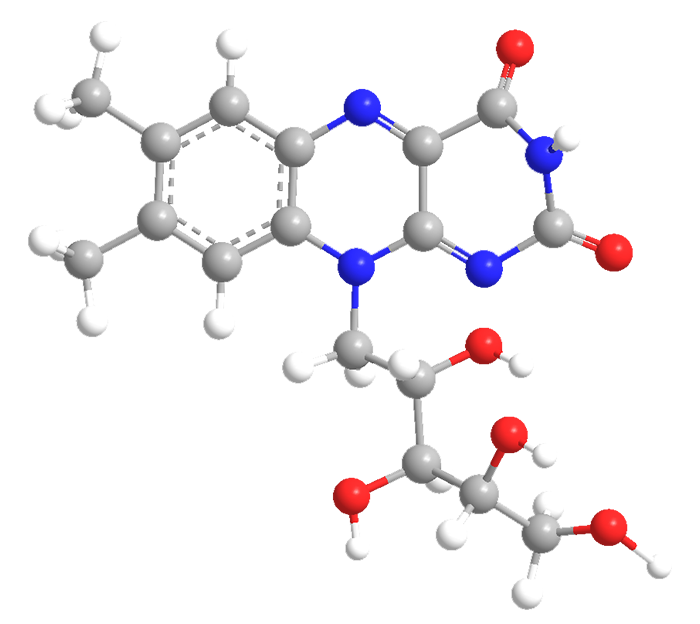
Riboflavin, also known as vitamin B2, is an enzyme cofactor in many flavoprotein enzyme reactions, notably the activation of other vitamins. It exists widely in nature, for example, in dairy products, eggs, organ tissues, and leafy vegetables. Yeast and its extracts are the richest natural source.
Riboflavin was first synthesized in Germany and Austria in the 1930s by chemists H. Meerwein and R. Kuhn and their colleagues. For use as a nutritional supplement, it was originally manufactured chemically, primarily from o-xylene, D-ribose, and alloxan. Nowadays it is biosynthesized with the use of fermenting organisms such as the fungus Ashbya gossypii and the bacterium Bacillus subtilis.
This year, J. B. Metternich and R. Gilmour at the University of Munster (Germany) found a new use for riboflavin. In a search for a better way to make Z-olefins, they noted that riboflavin can serve as a photocatalytic chromophore to isomerize the E-isomer of retinal (a vitamin A aldehyde found in retinas) to its Z-form. They used riboflavin to catalyze the selective, irreversible conversion of planar, conjugated E-olefins to their twisted, nonconjugated Z-counterparts. The researchers believe that this reaction will be useful in synthesizing complex molecules such as drugs and agrochemicals.
MOTW Update
The September 14, 2009, Molecule of the Week was paroxetine (trade name Paxil), an antidepressant. A recent study indicates that paroxetine’s mode of action may involve epigenetics. Some forms of depression involve enzymatic DNA methylation; paroxetine prevents the enzyme from being phosphorylated, an essential step in the enzyme’s activation.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

