What molecule am I?

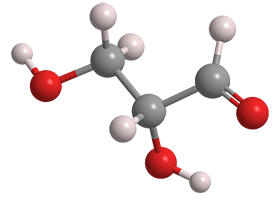
D-Glyceraldehyde, a triose and the simplest aldose (aldehyde sugar), is an intermediate in fructose metabolism. It occurs in all living organisms, including humans. Its enantiomer, L-glyceraldehyde1, is related to the natural amino acids; but it does not occur in nature.
Racemic glyceraldehyde, along with dihydroxyacetone2, can be synthesized via the hydrogen peroxide oxidation of glycerol. Further oxidation of rac-glyceraldehyde produces rac-glyceric acid3 and eventually one- and two-carbon compounds.
D-Glyceraldehyde is an important starting material for the classic Kiliani–Fischer synthesis, which is used to build larger sugars from smaller ones. In this synthesis, D-glyceraldehyde reacts with cyanide ion to add a carbon atom at the aldehyde position. The nitrile group is then hydrolyzed and catalytically hydrogenated to produce the tetroses D-erythrose4 and D-threose5. Further iterations of the Kiliani–Fischer synthesis lead to higher aldoses.
D-Glyceraldehyde is also involved in shortening aldose chains, as exemplified by the Wohl degradation. It essentially accomplishes the reverse of the Kiliani–Fischer synthesis, so that D-erythrose and D-threose can be degraded to D-glyceraldehyde in a three-step process that, like Kiliani–Fischer, proceeds through a nitrile intermediate.
For additional information, see the ScienceDirect topic page on D-glyceraldehyde. And a question for readers: Crystalline D-glyceraldehyde has a relatively high melting point of 145 °C—so why is the article of commerce a viscous syrup?
1. CAS Reg. No. 497-09-6.
2. CAS Reg. No. 96-26-4.
3. CAS Reg. No. 473-81-4.
4. CAS Reg. No. 583-50-6.
5. CAS Reg. No. 95-43-2.
D-Glyceraldehyde hazard information
| Hazard class* | GHS code and hazard statement |
|---|---|
| Not a hazardous substance or mixture** |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
**Most safety data sheets give this description; but one lists five hazard classes.
Molecule of the Future
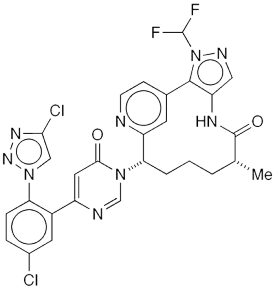
Milvexian1 is a developmental oral drug for preventing blood clots. It inhibits factor X1a (FX1a), an enzyme in the clot-forming coagulation cascade. Janssen Pharmaceuticals and Bristol-Myers Squibb (BMS) completed Phase 2 clinical trials on milvexian early this year.
In April, Simon Wagschal, Diego Broggini, and 35 colleagues at worldwide locations of Janssen and BMS reported a major advance toward the commercial synthesis of milvexian. They scaled up the reaction sequence for making the right-hand portion of the molecule (see image), which contains a 12-membered ring.
In June, Surasak Wichaiyo and co-workers at Mahidol University (Bangkok) reviewed the potential clinical benefits of small-molecule FXIa inhibitors for preventing arterial thrombosis. They focused on milvexian and another anticoagulant, asundexian2, along with mentions of other drug candidates in earlier stages of development. They concluded that small-molecule FXIa inhibitors have the advantages of rapid action, oral availability, and lower bleeding risks when compared with currently available anticoagulants.
1. CAS Reg. No. 1802425-99-5.
2. CAS Reg. No. 2064121-65-7.
Molecule of the Future
Once a month we bring you a newly discovered or developed molecule that has important implications for the future of chemistry or society in general. Look for it the third week of each month. Learn more about this month's Molecule of the Future below.
We're looking for more molecules of the future!
Do you have a suggestion for the next molecule of the future? Send your idea to MOTW.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
D-Glyceraldehyde fast facts
| CAS Reg. No. | 453-17-8 |
| SciFinder nomenclature | Propanal, 2,3-dihydroxy-, (2R)- |
| Empirical formula | C3H6O3 |
| Molar mass | 90.08 g/mol |
| Appearance | Colorless crystals or viscous liquid |
| Melting point | 145 °C |
| Water solubility | 29 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

