What molecule am I?

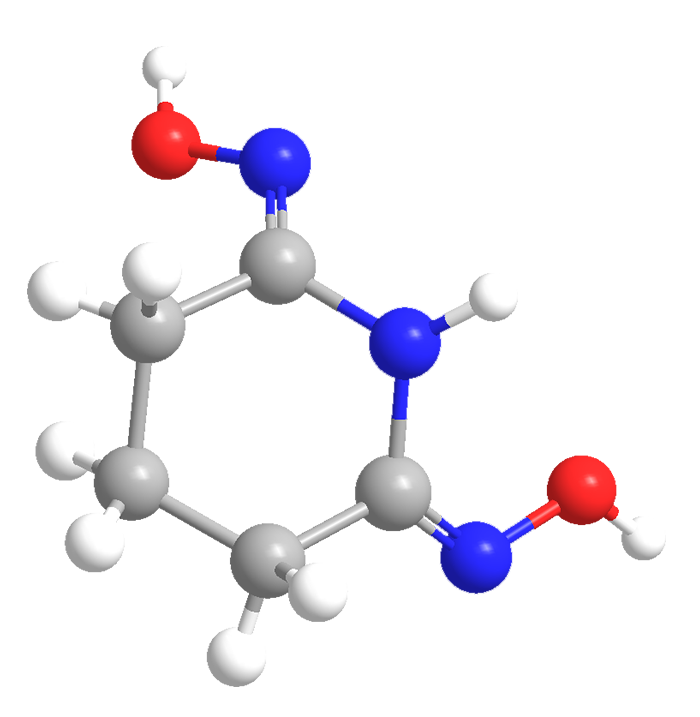
Glutarimide dioxime is a rather unusual compound with the ungainly systematic name 4,5-dihydro-6-(hydroxyamino)-2(3H)-pyridinone oxime. The trivial name arises because glutaric acid—a five-membered chain with a carboxyl on each end—reacts with a nitrogen source to form the heterocycle glutarimide. The imide carbonyl groups are then converted to oxime groups.
The main use for glutarimide dioxime is the complexation and removal of multivalent cations from water. Reports in recent years describe interactions between the ligand and common ions such as calcium and magnesium and more exotic ones such as platinum, lanthanides, and actinides.
About 6 months ago, Christina J. Leggett, Francesco Endrizzi, and Linfeng Rao* at Lawrence Berkeley National Laboratory (CA) initiated a series of articles in Industrial & Engineering Chemistry Research on the use of glutarimide dioxime to recover uranium from seawater. Their objectives were to find a way to supplement the world’s supply of nuclear fuel and eventually to replace terrestrial uranium mining.
It had previously been found that glutarimide dioxime forms very stable complexes with U(VI) even in the presence of the competing anion carbonate. The Berkeley researchers built on this finding by generating data that could be used to devise a practical uranium-recovery method.
The first article in the series covers the chemical speciation of uranium under seawater conditions. The second reports the results of fundamental thermodynamic and structural studies.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

