What molecule am I?

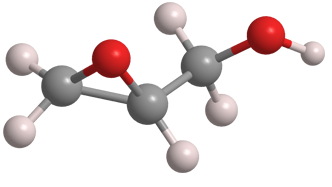
Glycidol is an epoxide and an alcohol, and as such is a highly reactive compound. It is miscible with water but also reacts with it. It decomposes when distilled at atmospheric pressure.
In 1909, chemist Nikolai Prilezhaev at the Warsaw University of Technology prepared glycidol by epoxidizing allyl alcohol with peroxybenzoic acid. This reaction was a breakthrough at the time and became known as the Prilezhaev oxidation. Glycidol is still manufactured in much the same way; but in 2018, a group of companies built a pilot plant in Teesside, UK, to make it via a “green” process.
Glycidol has a chiral center, but it is generally produced and used as the racemic mixture. It is used as a chemical intermediate and in the production of detergents, healthcare products, and industrial paints and coatings.
As shown in the hazard information table, glycidol presents an unusually large number of health risks. These factors were recently acknowledged when the US Food and Drug administration identified 19 substances as potentially harmful constituents of nicotine delivery systems (“e-cigarettes” and “e-liquids”). Glycidol, along with former Molecules of the Week diacetyl and ethylene glycol, is prominent in the FDA list. The agency now requires manufacturers and importers of the delivery systems to report the concentrations of each compound in its products.
Glycidol hazard information
| GHS classification*: flammable liquids, category 4 | |
| H227—Combustible liquid | |
| GHS classification: self-reactive substances and mixtures; and organic peroxides, type C | |
| H242—Heating may cause a fire | |
| GHS classification: acute toxicity, oral, category 4 | |
| H302—Harmful if swallowed | |
| GHS classification: acute toxicity, dermal, category 4 | |
| H312—Harmful in contact with skin | |
| GHS classification: skin corrosion/irritation, category 2 | |
| H315—Causes skin irritation | |
| GHS classification: serious eye damage/eye irritation, category 1 | |
| H318—Causes serious eye damage | |
| GHS classification: acute toxicity, inhalation, category 3 | |
| H331—Toxic if inhaled | |
| GHS classification: specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | |
| H335—May cause respiratory irritation | |
| GHS classification: germ cell mutagenicity, category 2 | |
| H341—Suspected of causing genetic defects | |
| GHS classification: carcinogenicity, category 1B | |
| H350—May cause cancer | |
| HS classification: reproductive toxicity, category 1B | |
| H360—May damage fertility or the unborn child | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Glycidol fast facts
| CAS Reg. No. | 556-52-5 |
| Empirical formula | C3H6O2 |
| Molar mass | 74.08 g/mol |
| Appearance | Colorless viscous liquid |
| Boiling point | 167 ºC (dec.) |
| Water solubility | Miscible |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

