What molecule am I?

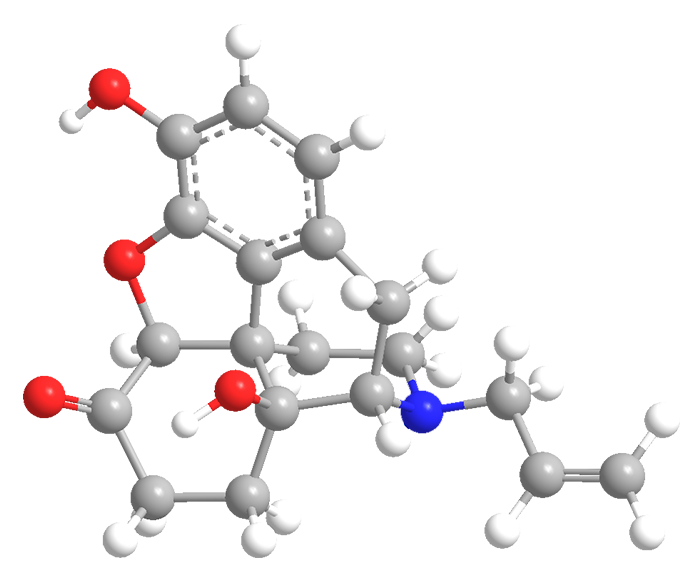
Naloxone—sold primarily under the trade name Narcan—has been widely publicized as a “rescue” drug for people whose lives are in danger from opioid overdoses. But this complex multiring compound has a long history.
Naloxone, a relative of morphine, has been recognized as an opiate antagonist since the early 1960s, when Mozes J. Lewenstein and Jack Fishman patented its synthesis in the United States, and the Japanese company Sankyo (now Daiichi Sankyo) patented it in Britain, among other countries.
In 1971, the US Food and Drug Administration approved it for treating opiate overdoses by intravenous or intramuscular injection. The moribund patient often begins breathing and regains consciousness immediately.
The rapid rise in prescription painkiller and heroin overdoses in the past decade spurred practitioners to seek ways other than injection to administer naloxone. At first, people improvised devices to deliver the drug nasally, but the results were often inadequate because of improper personnel training or inconsistent dosages.
In 2012, Phil Skolnick at the National Institute on Drug Abuse and Roger Crystal, CEO of Opiant Pharmaceuticals (formerly Lightlake Therapeutics) joined forces to develop an intranasal naloxone applicator that would be easy to use and would supply a known amount of drug. FDA approved the device in November 2015.
In February of this year, Opiant/Lightlake’s partner Adapt Pharma began to distribute Narcan nasal spray in the 38 states where it has been approved for use without a prescription. In the meantime, the Kaléo pharmaceutical company has begun to distribute Evzio, a new naloxone autoinjector.
MOTW update:
April 24, 2023
Naloxone1 (trade name Narcan) is an opiate antagonist first produced in the 1960s that is much in the news as a nasal-spray “rescue” drug for people who overdose on opioids. In late March, the US Food and Drug Administration approved naloxone for over-the-counter sales. Its manufacturer says that it should be available in stores and online by late summer.
1. CAS Reg. No. 465-65-6.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

