What molecule am I?

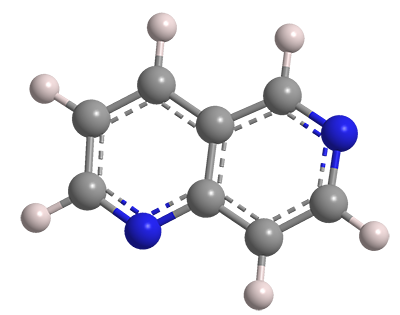
Naphthalene is an aromatic organic compound that consists of two fused benzene rings. Diazanaphthalenes have the naphthalene structure with nitrogen atoms in place of two of the CH groups. There are two structural classes of diazanaphthalenes: benzodiazines, in which both nitrogen atoms are in the same ring, and naphthyridines, with one nitrogen atom in each ring.
Six naphthyridine isomers exist, based on the positions of the nitrogen atoms; they can be in the 1,5, 1,6 (shown here), 1,7, 1,8, 2,6, or 2,7 positions. All are white solids with a surprisingly wide span of melting points: 1,6-Naphthyridine’s is the lowest at <40 ºC; 2,6-naphthyridine’s is the highest at 114–115 ºC.
Quinoline derivatives such as naphthyridines that have nitrogen atoms in the 1-position are usually synthesized via the Skraup reaction, in which an aminopyridine and a glycerol derivative are heated in the presence of an oxidizing agent (e.g., nitrobenzene) and sulfuric acid. This is often a violent, runaway reaction, so it is perhaps fortunate that the synthesis of 1,6-naphthyridine from 4-aminopyridine was not originally successful.
Refinements of the Skraup reaction, however, did eventually lead to preparations of 1,6-naphthyridine in modest yields. One modification used 4-aminopyridine-N-oxide as the starting material; the resulting 1,6-naphthyridine-N-oxide was then reduced to the free base.
1,6-Naphthyridine and some of its derivatives have been reported to have medicinal, electronic, and catalytic properties. But none of these investigations has yet resulted in any practical applications.
1,6-Naphthyridine hazard information
| Hazard class*,** | Hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 4 | H302—Harmful if swallowed | |
| Serious eye damage/eye irritation, category 1 | H318—Causes serious eye damage | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
**Some safety data sheets classify as “not a hazardous substance or mixture”.
MOTW update
1,4-Dioxane was the Molecule of the Week for August 5, 2019. Dioxane is a valuable solvent that has many industrial and laboratory uses. In 2019, it came under regulatory pressure because it is a possible carcinogen and has been found in drinking water and groundwater in several states. On December 31, 2020, the US Environmental Protection Agency released a hastily compiled risk assessment of dioxane that pleased neither environmentalists, state attorneys general, nor the chemical industry. Among the report’s other shortcomings, EPA ignored drinking-water exposure; environmentalists and regulators say that this is dioxane’s greatest threat to the general population.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
1,6-Naphthyridine
fast facts
| CAS Reg. No. | 253-72-5 |
| SciFinder nomenclature | 1,6-Naphthyridine |
| Empirical formula | C8H6N2 |
| Molar mass | 130.15 g/mol |
| Appearance | White to pale yellow or light brown solid or liquid |
| Melting point | 31–38 ºCa |
| Water solubility | Unreported |
a. Wide range of values reported.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

