What molecule am I?
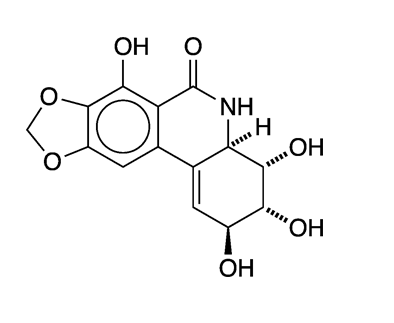
Narciclasine, also known as lycoricidinol, is an isocarbostyril alkaloid found in the Amaryllidaceae (amaryllis) family of flowering plants. One notable genus of the family is Narcissus, from which narciclasine gets its name. Daffodils and jonquils are members of the Narcissus genus.
In 1967, Giovanni Ceriotti, Luigi Spandrio, and Annivale Gazzaniga at Circolo Hospital (Busto Arsizio, Italy) isolated narciclasine from Narcissus varieties. At that time, it was known to inhibit cell division. By the early 2010s, narciclasine was an established antitumor agent.
In 2016, Robert Fürst at Goethe University (Frankfurt am Main, Germany) reviewed the antitumor and anti-inflammatory findings on narciclasine. At present, no clinical trials on narciclasine have been conducted on humans.
Narciclasine is sparingly soluble in water. But in 2003, George R. Petit and colleagues at Arizona State University (Tempe) synthesized a cyclic phosphate at two of the adjacent hydroxyl groups of narciclasine. The product, which they called narcistatin (CAS Reg. no. 496963-44-1), is much more water-soluble (4 g/L), making it a valuable prodrug for narciclasine.
Narciclasine hazard information
| GHS classification*: germ cell mutagenicity, category 1B | |
| H340—May cause genetic defects | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Narciclasine fast facts
| CAS Reg. No. | 29477-83-6 |
| Molar mass | 307.26 g/mol |
| Empirical formula | C14H13NO7 |
| Appearance | White to off-white powder |
| Melting point | 246 ºC |
| Water solubility | Slight |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

