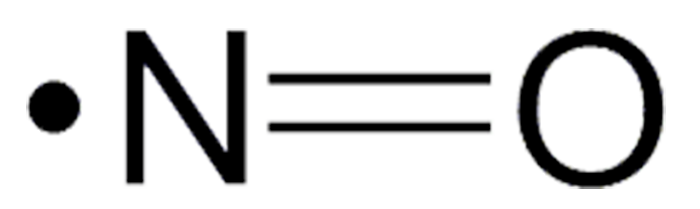
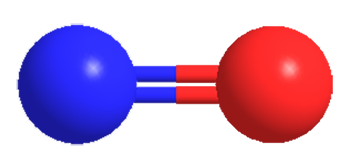
Nitric oxide (NO) is a colorless gas and stable free radical that turns blue when it is liquefied or solidified. It was discovered and studied in 1772 by J. Priestley, who called it “nitrous air”. A toxic gas and air pollutant, NO has many industrial uses, especially in the production of nitric acid.
In the 1980s it was discovered that NO is the active metabolite released from nitroglycerine and amyl nitrite, vasodilators that are used for treating heart conditions such as angina. R. F. Furchgott, L. J. Ignarro, and F. Murad won the 1998 Nobel Prize in Physiology or Medicine for their work on NO’s role as a physiological signaling molecule. NO is released by the noni plant (Morinda citrifolia), which has been used as a “cure-all” by Pacific islanders for thousands of years.
MOTW update: June 1, 2020
Nitric oxide (NO) was the Molecule of the Week for February 4. 2013 . NO is a toxic gas; but in the body, it has an important role as a signaling molecule. Currently, it’s being evaluated as a treatment for COVID-19. During the 2003 SARS epidemic, Patients treated with NO recovered faster than patients who were not treated. At least 11 NO clinical trials are under way on COVID-19 patients.
MOTW update:
January 23, 2023
Nitric oxide1 (NO) is a colorless, toxic gas that is a stable free radical and a physiological signaling molecule. This month, Ke Chu and coauthors at Lanzhou Jiaotong University (Lanzhou) and Henan University (Kaifeng, both in China) reported that NO can be electrolytically hydrogenated to ammonia on molybdenum carbide (Mo2C) nanosheets, a catalyst that selectively activates NO.
1. CAS Reg. No. 10102-43-9.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

