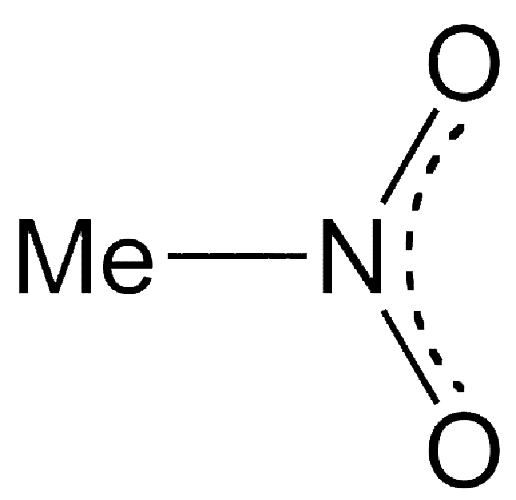
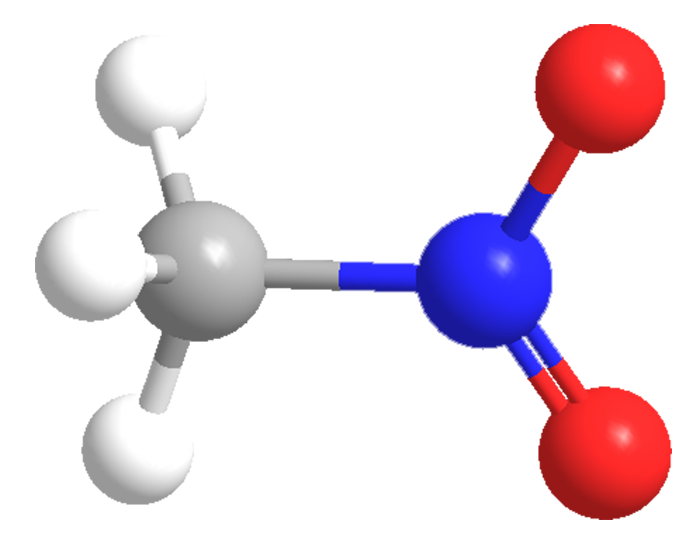
Nitromethane is a strongly polar, oily liquid that is used as a solvent or reactant in organic chemical synthesis and as a fuel in motor sports, rockets, hobby airplanes, and the like. It is produced, along with other nitroalkanes, in a high-temperature reaction between propane and nitric acid.
Drag racers such as John Force and his family use nitromethane as a fuel because it requires only about one-ninth as much air as gasoline for complete combustion. Although it has only about one-fourth the combustion energy of gasoline, its lower air requirement gives it the advantage of producing the high instantaneous thrust needed for drag racing.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

