On October 5, the Nobel Committee awarded the 2016 prize in chemistry to Jean-Pierre Sauvage of the University of Strasbourg (France), J. Fraser Stoddart of Northwestern University (Evanston, IL), and Ben L. Feringa of the University of Groningen (The Netherlands). In the 1980s, these researchers pioneered work that led to the creation of molecular machines—molecules that can perform work when they are energized.
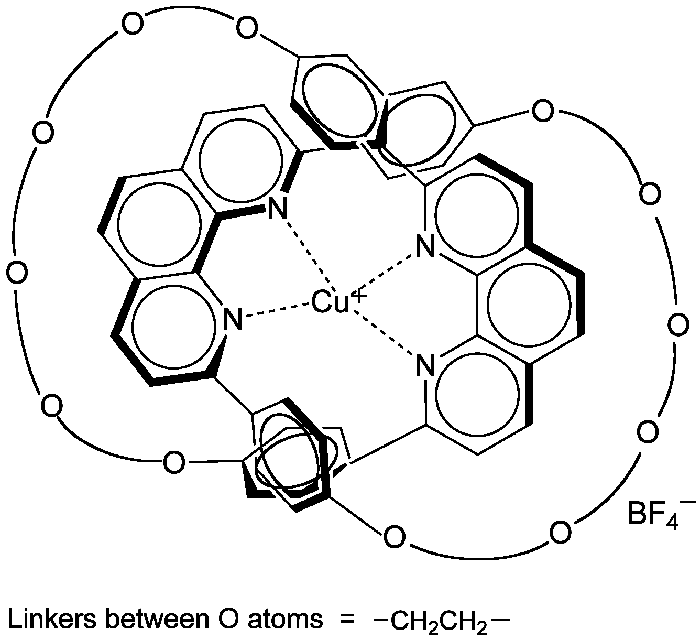
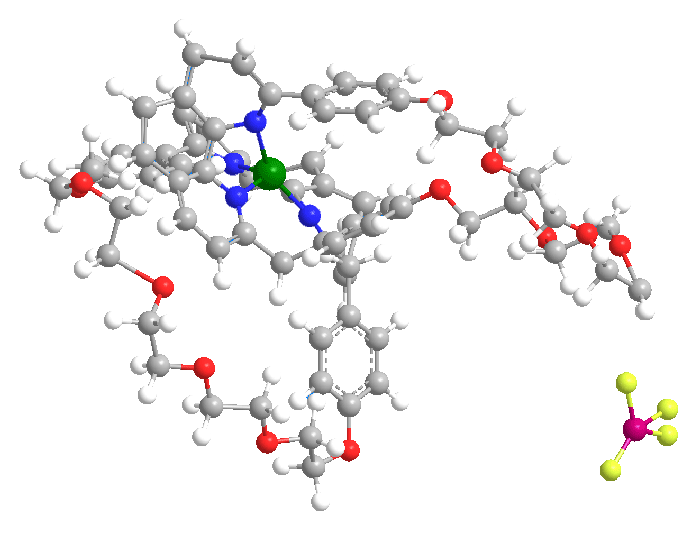
Sauvage and co-workers described the molecule shown here in a 1983 paper. The structure consists of a functionalized catenane coordinated to a copper(I) ion. (Catenanes are molecules that contain ring structures that are linked but not connected by a covalent bond.)
The authors reported that the molecule was the first example of a metallo-catenane. They also stated that their synthetic method was analogous to the action of the enzyme gyrase, which uses a catenation process to relieve strain when double-stranded DNA is unwound by another enzyme.
Molecular motors pioneered by Feringa in 1999 relied on catenanes developed by Sauvage and rotaxanes synthesized by Stoddart. A rotaxane is similar to a catenane in that it consists of two pieces that are not covalently connected. The difference is that rotaxanes have only one ring, through which is threaded a linear, dumbbell-shaped structure that can’t escape from the ring.
MOTW Update
Vitamin A was one of the Molecules of the Week for June 9, 2014. This week’s Chemical & Engineering News reports that BASF plans to build a 1500-t/year vitamin A acetate plant that will open in 2020. The acetate is the article of commerce because it is more stable than the free vitamin A alcohol.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

