What molecule am I?

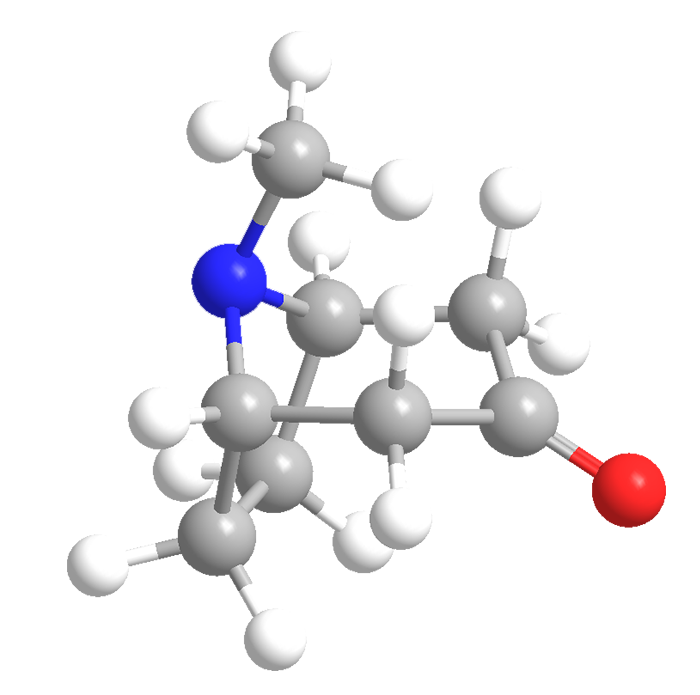
Since the 4th century BCE, Mandragora spp. (mandrake), Atropa belladonna (deadly nightshade), and other plants in the Solanaceae family were known to have medicinal uses—and to be poisonous. By the 19th century, scientists had discovered that one of the most important bioactive components of these plants was atropine. It was prepared in pure form by pharmacist Heinrich F. G. Mein in 1831 and synthesized by chemist Richard Willstätter in 1901.
Willstätter synthesized atropine from tropine and tropic acid, both of which must be made separately from available materials. The alcohol tropine can be prepared from its ketone form tropinone, which at the time was difficult to synthesize. Willstätter’s convoluted synthesis of tropinone from cycloheptanone required 15 steps and had an overall yield of 0.75%.
When atropine was needed to overcome the effects of nerve gases during World War I, very little was available. That’s when Britain’s Sir Robert Robinson developed his classic one-pot, concerted synthesis of tropinone from succinaldehyde, methylamine, and acetonedicarboxylic acid. The original yield was 17%; it was later improved to an astounding 90%.
To this day, tropinone’s only practical use is as a precursor to atropine. Robinson was awarded the 1947 Nobel Prize in Chemistry—however, not for his tropinone synthesis.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

