What molecule am I?

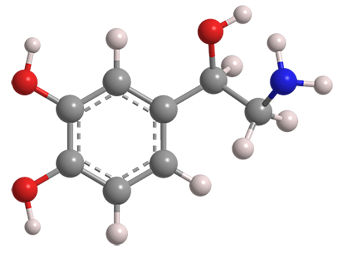
Norepinephrine is a natural compound in the catecholamine family that acts as a hormone and neurotransmitter. Its original name was noradrenaline, which is still used in the United Kingdom. The United States and the World Health Organization use norepinephrine. “Nor” in norepinephrine indicates that it has the structure of the parent molecule epinephrine with a hydrogen atom in place of the N-methyl group.
According to the Merck Index, norepinephrine “occurs in animals and [humans] and is a sympathomimetic hormone of both adrenal origin and adrenergic orthosympathetic postganglionic origin in [humans].” As an adrenal hormone, its release triggers vasoconstriction, which increases blood pressure. Despite its hazards (see table), this characteristic led scientists to develop norepinephrine as an intravenous drug for treating patients with very low blood pressure.
Physiologist Ulf van Euler at the Karolinska Institute (Solna, Sweden) discovered norepinephrine’s role in the body in 1945. He shared the 1970 Nobel Prize in Physiology or Medicine for his work on neurotransmitters.
Earlier this year, researchers reported a previously unknown property of norepinephrine. Ya-Chieh Hsu and colleagues at Harvard University (Cambridge, MA) and other research institutions used laboratory mice to determine the underlying biochemical cause of stress-induced hair depigmentation. They found that the culprit is norepinephrine, which is released by mammals’ adrenal glands when animals are under stress.
But the authors’ big surprise came when they removed the adrenal glands from mice to test their finding: The rodents still went gray. Undaunted, the researchers discovered that animals under stress also release norepinephrine from the sympathetic nervous system. The chemical reaches hair follicles and depletes them of pigment-producing stem cells.
Norepinephrine hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Acute toxicity, oral, category 2 | H300—Fatal if swallowed | |
| Acute toxicity, dermal, category 2 | H310—Fatal in contact with skin | |
| Acute toxicity, inhalation, category 1 | H330—Fatal if inhaled | |
*Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Norepinephrine
fast facts
| CAS Reg. No. | 51-41-2 |
| SciFinder nomenclature | 1,2-Benzenediol, 4-[(1R)-2-amino-1-hydroxyethyl]- |
| Empirical formula | C8H11NO3 |
| Molar mass | 169.18 g/mol |
| Appearance | Off-white to tan crystals; colorless microcrystals |
| Melting point | 217 ºC (dec) |
| Water solubility | 0.85 g/L |
MOTW Update
Ethylene oxide was the Molecule of the Week for November 18, 2019. It is used extensively in chemical manufacturing, agriculture, and fumigation, but it is extremely hazardous. In March 2020, the US Environmental Protection Agency’s Office of the Inspector General found that the agency had not done enough to inform people who are living near ethylene oxide–emitting chemical plants that they are likely to have an increased risk of cancer. The OIG even recommended that EPA use unverified data to warn neighbors of the hazard.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

