What molecule am I?
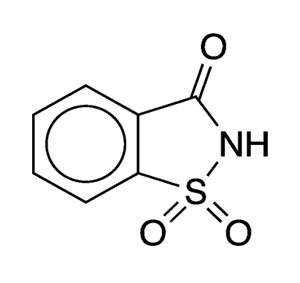
Saccharin was the first widely commercialized non-nutritive sweetener. It was discovered in 1879 by Constantin Fahlberg in the laboratory of Ira Remsen at Johns Hopkins University (Baltimore).
Fahlberg was working with the coal tar derivative benzoic sulfimide when he noticed a sweet taste on his hand.1 He connected the taste with the compound, which he gave the trivial name saccharin. He and Remsen soon developed a synthesis of saccharin from o-sulfamoylbenzoic acid. In the mid-1880s, Fahlberg, much to Remsen’s distress, began to manufacture saccharin in Germany.
As early as 1906, saccharin became controversial because of food additive concerns prompted by Upton Sinclair's The Jungle. Harvey Wiley, the head chemist at the US Department of Agriculture, proposed a saccharin ban; but President Theodore Roosevelt, who needed to lose weight, declared, “Anyone who says saccharin is injurious to health is an idiot.” Wiley’s career was finished.
Saccharin’s use became widespread during World War I because of a sugar shortage. In the 1960s, it began to be promoted for weight loss, most familiarly under the trade name Sweet’n Low (Cumberland Packing Corp., Brooklyn).
Soon thereafter, food scientists discovered that saccharin causes bladder cancer in rats. In 1977 an act of Congress required the sweetener’s packaging to bear a cancer warning label. But in 2000, scientists found that humans metabolize saccharin differently from rats. The warning label requirement was rescinded.
Today, despite all the competition from other sweeteners and its metallic aftertaste, saccharin remains a popular choice.
1. In another version of the discovery, Fahlberg carelessly laid his cigarette on his lab bench. When he next smoked it, the cigarette tasted sweet.
Saccharin hazard information
| GHS classification*: not a hazardous substance or mixture |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Saccharin fast facts
| CAS Reg. No. | 81-07-2 |
| Empirical formula | C7H5NO3S |
| Molar mass | 183.18 g/mol |
| Appearance | White crystals |
| Melting point | 229–230 ºC |
| Water solubility | 3.45 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.


