What molecules are we?
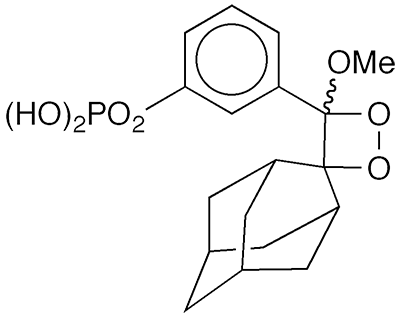
In the late 1980s, A. Paul Schaap and co-workers at Wayne State University (Detroit) reported the synthesis of dioxetane derivatives that trigger bright chemiluminescence when activated by alkaline phosphatase enzymes (see image). These compounds came to be known as “Schaap’s dioxetanes”.
One of Schaap’s compounds, 3-(2′-spiroadamantane)-4-methoxy-4-(3″-phosphoryloxy)phenyl-1,2-dioxetane1—known as PPD marketed by Lumigen2 (Southfield, MI)—is of particular interest. It is stable at ambient temperature; and in 1989, Schaap*, Hashem Akhavan, and Louis J. Romano reported that it is suitable for ultrasensitive enzyme-linked immunoassays and DNA probes. The same year, Irena Bronstein and co-workers at Tropix3 (Bedford, MA) described its use in immunoassays of the thyroid-stimulating hormone thyrotropin. Typically, PPD’s more stable disodium salt is used in immunoassays rather than the free acid.
PPD is used in more than 2 billion chemiluminescent immunoassays per year for clinical diagnosis, food safety, pharmaceutical analysis, and environmental monitoring. In 2018, Roland Lindh at Uppsala University (Sweden) and Harvard University (Cambridge, MA) and coauthors around the world wrote a comprehensive review of the chemiluminescence and bioluminescence of cyclic peroxides, including Schaap’s dioxetanes. The following year, Nir Hananya and Doron Shabat*4 at Tel Aviv University reported that adding an electron-withdrawing group to one of the dioxetanes increased its chemiluminescence quantum yield by 3000-fold.
1. SciFinder: phenol, 3-(4-methoxyspiro[1,2-dioxetane-3,2′-tricyclo[3.3.1.13,7]decan]-4-yl)-, 1-(dihydrogen phosphate).
2. Now part of Beckman Coulter (Brea, CA).
3. Now part of PerkinElmer (Waltham, MA).
4. Shabat was one of the early coiners of “Schaap’s dioxetanes”.
PPD hazard information
| Hazard class* | GHS code and hazard statement |
|---|---|
| Not a hazardous substance or mixture |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
Molecules from the journals
Pentachloronitrobenzene1 (PCNB), also called quintozene, was first synthesized in 1868 by Emil Jungfleisch at the École Supérieure de Pharmacie (Paris) via the reaction of pentachlorobenzene with fuming nitric acid. Its use as a pesticide began in the 1930s; it is marketed as a fungicide for turfgrass and some food crops. Because of its hazards to humans and the environment, the US Environmental Protection Agency proposed a ban on all uses of PCNB in September 2022.
Pterostilbene2 (trans-3,5-dimethoxy-4-hydroxystilbene) is a natural product related to trans-resveratrol3; it is found in almonds, some berries, and grape leaves and vines. It acts as an antimicrobial in plants; but in concentrated form, it is an eye hazard and harmful to aquatic environments. In October 2022, Hao Zhang, Tian Wang, and colleagues at Nanjing Agricultural University (China) and Jiangsu Academy of Agricultural Sciences (Nanjing) reported that pterostilbene prevents tunicamycin-induced intestinal barrier damage by targeting endoplasmic reticulum stress, oxidative stress, autophagy, and gut microbiota in piglets.
1. CAS Reg. No. 82-68-8.
2. CAS Reg. No. 537-42-8.
3. CAS Reg. No. 501-36-0.

Molecules from the Journals
MOTW briefly describes noteworthy molecules that appeared in recent ACS journal articles. See this week's
edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
PPD fast facts
| CAS Reg. No. | 122341-56-4 |
| Empirical formula | C18H23O7P |
| Molar mass | 382.34 g/mol |
| Appearance | White crystals |
| Melting point | ≈250 °C (dec., luminescence) |
| Water solubility | 630 g/L (est.) |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

