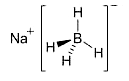
Sodium borohydride (NaBH4) is one of the handiest reducing agents, especially for organic compounds in nonaqueous solvents. It was introduced in the 1940s for wartime applications and was an important enough reagent by 1970 to warrant a lengthy review. It has been used to reduce aldehydes, ketones, Schiff bases, carboxylic acids and esters, acid chlorides, disulfides, nitriles, and inorganic anions. For many of these substrates, only gentle reaction conditions are required.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

