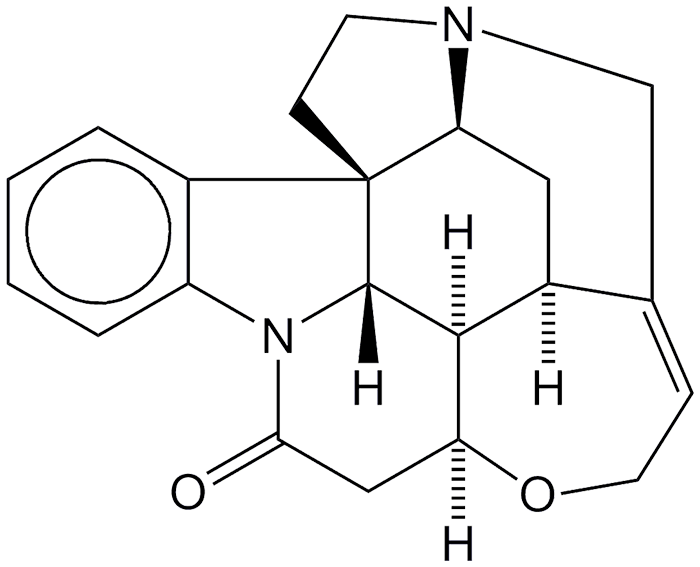
Strychnine is an extremely poisonous alkaloid produced in the seeds of trees and shrubs of the Strychnos genus; it was identified and named by C. Linnaeus in 1753. J. B. Caventou and P.-J. Pelletier discovered it in Saint Ignatius's bean (from the Strychnos ignatii shrub) in 1818.
R. Robinson determined the structure of strychnine in 1946; and R. B. Woodward and co-workers completed a classic total synthesis in 1954. Both chemists were awarded the Nobel Prize in Chemistry for their work in 1947 and 1965, respectively.
Strychnos seeds and strychnine were used for centuries to kill animal and bird pests, but this use is no longer permitted. Strychnine has been implicated in many murders and attempted murders, possibly dating back to Alexander the Great.
Recently, J. R. Cort and A. E. Metaxas at Pacific Northwest National Laboratory developed an NMR method for distinguishing strychnine salts. If their method can be extended to other poisons and drugs, it could prove useful in forensic chemistry.
MOTW update:
July 4, 2022
(–)-Strychnine1 is a virulent poison produced in the seeds of plants of the Strychnos genus. Its non-natural enantiomer, (+)-strychnine2, was first synthesized in 1995.
In the 1940s, Robert Robinson (Oxford University) and Robert B. Woodward (Harvard University), both eventual Nobel laureates in chemistry, published structures of the natural strychnine molecule. Since then, there has been controversy as to which chemist should be credited for first describing the true structure.
In a recent survey of chemists conducted by Jeffrey I. Seeman* at the University of Richmond (VA) and Mark C. House at Santa Fe College (Gainesville, FL), the respondents did not reach a consensus as to which Nobelist deserves the credit. The authors concluded that
1. CAS Reg. No. 57-24-9.
2. CAS Reg. No. 163956-38-5.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

