What molecule am I?
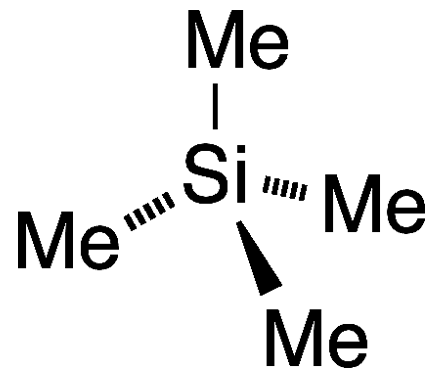
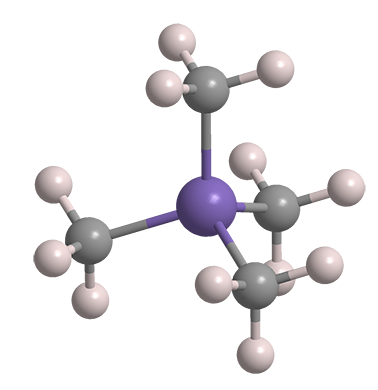
Tetramethylsilane (TMS1) is the simplest tetraalkylsilane and the organosilicon analogue of neopentane. It is a volatile liquid with a characteristic “organometallic” odor.
TMS has been known since at least 1911, when Artur Bygden at the University of Uppsala (Sweden) prepared it from silicon tetrachloride and the Grignard reagent2 methylmagnesium chloride. Nowadays, TMS is made less expensively, as one of the products of the direct alkylation of elemental silicon with chloromethane.
TMS is sometimes used as a starting material for synthesizing more complex organosilanes, but its main use by far is as a versatile internal standard in NMR spectroscopy. Its 1H, 13C, and 29Si signals are sharp and have resonances that are distinctly different from the nuclei normally encountered in test samples.
For example, the 1H atoms in TMS are highly shielded by the silicon atom and therefore resonate at a higher magnetic field than 1H atoms surrounded by more common nuclei such as carbon, oxygen, and nitrogen. TMS is such a common standard that its 1H chemical shift (δ) is defined as zero. In most 1H spectra, all of the signals are to the left (lower field) of the TMS signal, although highly shielded protons in some aromatic compounds can appear to the right of TMS.
Highly soluble TMS is an ideal standard for generating NMR spectra of samples in organic solvents, but its low water solubility makes it unusable for aqueous samples. TMS’ low cost, chemical inertness, and ease of removal because of its volatility are additional factors that make it the universal standard for organic-solution NMR.
1. TMS is also used as the abbreviation for the trimethylsilyl radical.
2. Victor Grignard, the French Nobel Prize–winning chemist, had recently discovered the eponymous reaction; and chemists around the world were experimenting with alkylmagnesium halides in all sorts of ways.
Tetramethylsilane hazard information
| Hazard class* | Hazard statement | |
|---|---|---|
| Flammable liquids, category 1 | H224—Extremely flammable liquid and vapor | |
| Hazardous to the aquatic environment, acute hazard, category 2 | H401—Toxic to aquatic life | |
*Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Tetramethylsilane
fast facts
| CAS Reg. No. | 75-76-3 |
| SciFinder nomenclature | Silane, tetramethyl- |
| Empirical formula | C4H12Si |
| Molar mass | 88.22 g/mol |
| Appearance | Colorless liquid |
| Boiling point | 26.6 ºC |
| Water solubility | 20 mg/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

