What molecule am I?
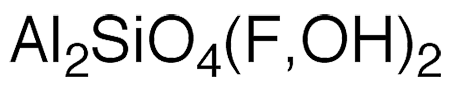
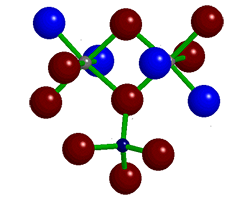
Topaz is an aluminosilicate with varying amounts of fluoride and hydroxide ions. The 3-D image (shown) is a portion of its crystal unit cell in which the small gray spheres are aluminum, the small black sphere is silicon, the large red spheres are oxygen, and the large blue spheres can be fluorine or hydroxide. A naturally occurring gemstone, topaz is the birthstone for November.
Pure crystalline topaz is colorless; but impurities in the natural stone cause it to appear in a wide range of colors. Common varieties are yellow to brown (iron), blue (chromium or titanium), and pink to red (manganese). Topaz is one of the hardest natural materials, with a rating of 8 on the 1–10 Mohs scale in which diamond is defined as 10.
A very early mention of topaz in the chemical literature appeared in 1881 when geologist Alexis A. Julien at Columbia University (New York City) wrote an extensive treatise on carbon dioxide in the fluid cavities of the mineral. Julien describes the occurrence of CO2 in topaz samples from several locations worldwide; but he does not seem to arrive at any definite conclusions.
More than a century later, M. S. Hampar and J. Zussman at the University of Manchester (UK) described the thermal decomposition of topaz. With the use of X-ray diffraction, thermal analysis, and electron microscopy, they found that at ≈1000 °C topaz begins to form silica (SiO2) and mullite [(Al2O3)3–4(SiO2)2]. The authors noted that the presence of fluoride reduced the decomposition point of the silica–alumina system, which is normally in the range of 1700–1800 °C.
Topaz hazard information
| Hazard class* | GHS code and hazard statement |
|---|---|
| Not classified; most likely not a hazardous substance |
*Globally Harmonized System (GHS) of Classification and Labeling of Chemicals.
Molecules in the News
Tetrachlorvinphos1 (TCVP) is a polychlorinated organophosphate insecticide that was developed in the 1960s by Shell Development Co. (then in Modesto, CA) for use on food crops, animals, and places of occupancy. Crop use was canceled in 1987; today, it is used primarily for flea and tick control on pets.
In 2022, the US Environmental Protection Agency banned the use of TCVP in pet collars, citing that children could be exposed to dangerous levels of it from animal fur. This past September, however, EPA reversed its decision based on data submitted by Hartz Mountain Corp. (Secaucus, NJ), which manufactures pet collars that contain TCVP.
Ubiquinol2 and ubiquinone3 are the reduced and oxidized forms, respectively, of coenzyme Q10 (CoQ10), which is important for cell growth and maintenance in animals and bacteria. Both forms are sold over the counter as dietary supplements for replacing age-related CoQ10 decline and reducing statin-associated muscle pain and weakness. Ubiquinol has greater bioavailability than ubiquinone but is subject to air oxidation if it is not stabilized.
Kaneka Corp. (Osaka, Japan) developed and patented a way to stabilize ubiquinol from oxidation; the company has been selling it in the United States since 2007. Kaneka claims that it has the only legal right to sell ubiquinol in the United States. Last month, it sued Cocrystal Technology and Cocrystal Health Industry (Jiaxing, China) for infringing its patent. Kaneka alleges that Cocrystal-produced CoQ10 samples obtained from a US importer contained 93% ubiquinol.
1. CAS Reg. No. 22248-79-9.
2. CAS Reg. No. 5677-55-4.
3. CAS Reg. No. 606-06-4.
Molecules in the News
MOTW highlights molecules that appear in major news outlets. See this week's edition below.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
Topaz fast facts
| CAS Reg. No. | 1302-59-6 |
| SciFinder nomenclature | Topaz {Al2[F0.5–1 (OH)0–0.5]2 SiO4} |
| Empirical formula | Al2F1–2H0–1 O4–5Si |
| Formula mass | 182.05–184.04 g/mol |
| Appearance | Colorless crystalsa |
| Melting point | >1000 °C (dec.) |
| Water solubility | Insolubleb |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

