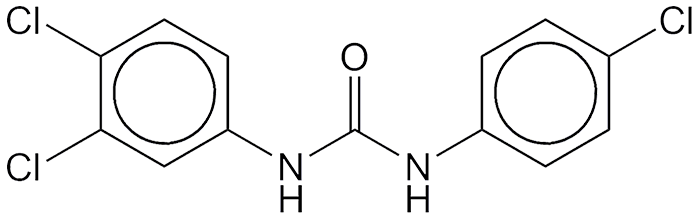
Triclocarban, along with its cousin triclosan, is an antibacterial and antifungal component of personal care products such as soaps, toothpaste, and deodorants. It has been in use since the 1960s, but it has recently come under fire from the US Food and Drug Administration.
A rule proposed by the FDA in December 2013 would require manufacturers of antibacterial soaps and body washes to demonstrate that their products are safe for everyday use and are more effective than products without antibacterials. Proponents of the rule argue that users' risk of bacterial infection is low; and therefore, the antibacterial additives are not needed. Producers rebut this argument by citing dozens of studies that show that antibacterial soaps significantly reduce bacteria levels on skin compared with ordinary soaps.
More recently, a bill in the New York State assembly would, if passed, ban the sale of antibacterial personal care products in that state.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

