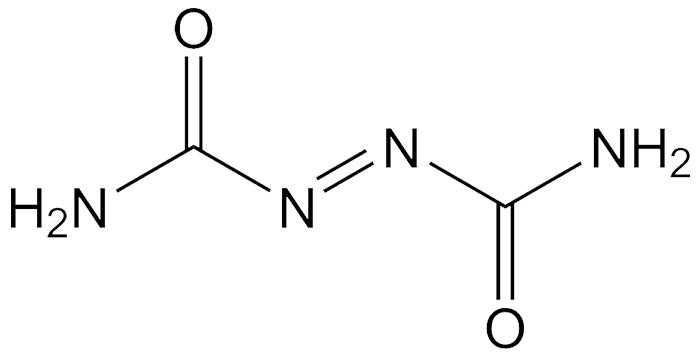
Azodicarbonamide, the diamide of azodicarboxylic acid, is an orange-red crystalline solid. It is made industrially by the condensation reaction between hydrazine sulfate and urea under high temperature and pressure, followed by oxidation with NaOCl.
Azodicarbonamide has several commercial uses: It is a blowing agent for foaming rubbers and plastics, a bleaching (oxidizing) agent in cereal flours, and a dough conditioner for baking bread.
In the United States, azodicarbonamide has GRAS (generally recognized as safe) status for food use. The World Health Organization also says it’s safe. But European countries and Australia have banned its use in food products, and some European countries have outlawed it for making plastics that come into contact with food. Some studies suggest that consuming it or its byproducts may lead to respiratory problems.
Earlier this year, the Subway fast-food chain, calling it a “shoe rubber chemical”, decided to remove azodicarbonamide from its bread-making process.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

