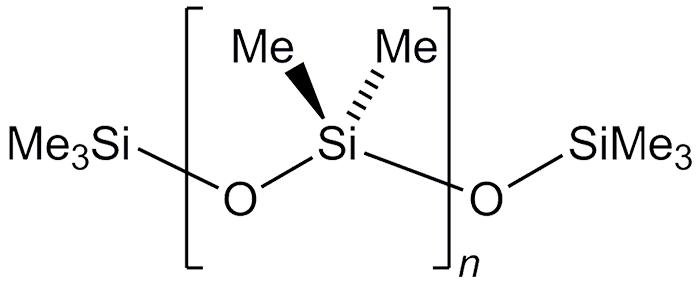
Polydimethylsiloxane (PDMS) is the simplest member of the silicone polymer family. It is formed by hydrolyzing Me2SiCl2, which is produced from high-purity SiO2 and CH2Cl2 by the Muller–Rochow reaction. The term “silicone” was coined by chemist F. S. Kipping in 1901.
Low–molecular weight PDMS is a liquid used in lubricants, antifoaming agents, and hydraulic fluids. Its use in breast implants is not as popular as it once was because of safety concerns.
At higher molecular weights, PDMS is a soft, compliant rubber or resin. It is used in caulks, sealants, an even Silly Putty. More recently, PDMS resins have been used in soft lithography, a key process in biomedical microelectromechanical systems (bio-MEMS).

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

