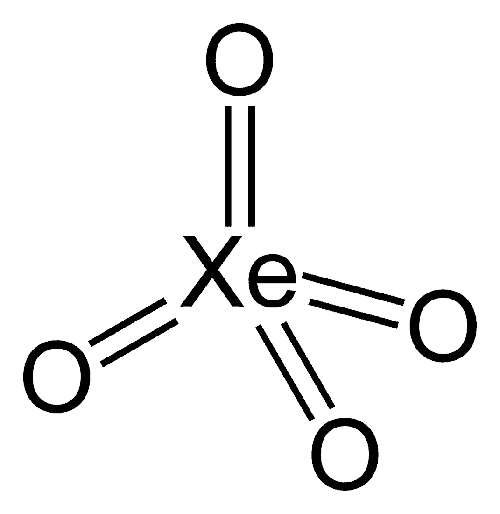
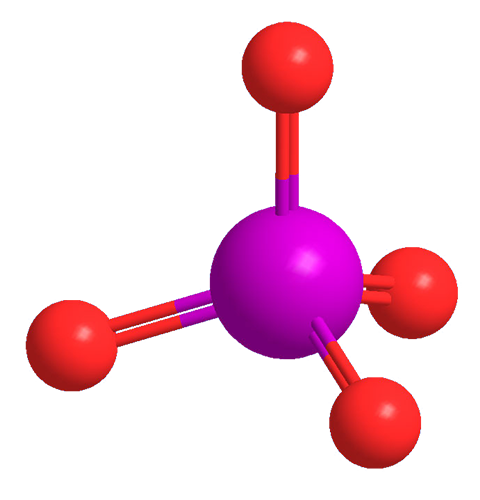
Xenon tetroxide (XeO4) is an unusual noble gas compound. It contains xenon in its highest possible oxidation state (+8). Its yellow crystals are stable below –36 °C, but it decomposes explosively above that temperature. It must be handled under strict safety precautions. XeO4’s tetrahedral geometry follows valence shell electron pair repulsion (VSEPR) rules.

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

