What molecules are we?
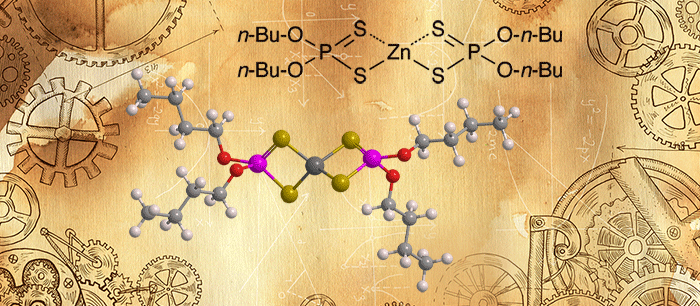
Zinc dialkyl dithiophosphates (ZDDPs) are very effective antiwear agents that are used as machine lubricants (e.g., greases, hydraulic oils, and motor oils), including applications in the automotive industry.
According to a 2004 article by Hugh Spikes at Imperial College London, ZDDPs were developed in the 1940s. Today, they are still by far the most prevalent antiwear additive in heavy equipment and off-road vehicle engine oils. They are not used in passenger vehicles because the phosphorus in ZDDPs prematurely disables catalytic converters in exhaust systems.
On paper, ZDDPs look like zinc salts, but their properties much more closely resemble those of organic compounds. As an example, they are soluble in nonpolar organic solvents, including oils, and are insoluble in water.
Zinc dibutyl dithiophosphate, shown in the images and information tables, is a typical ZDDP. But commercial ZDDPs almost always contain a mixtures of alkyl groups, ranging from C4 to C12. The specific mixture used depends on the type of lubricant and metallurgy involved. Shorter-chain alkyl groups, however, are the least thermally stable; they thus make protective films more easily.
Currently, concerns over ZDDPs’ environmental effects are driving efforts to curtail or eliminate their use. But no one has found a substitute that performs quite as well; and how harmful ZDDPs actually are at the level of usage is the topic of some rather heated discussions among lubricant formulators, vintage car enthusiasts, and environmental advocacy groups.
[Many thanks to Nancy McGuire for assistance with this article—ED].
Zinc dibutyl dithiophosphate hazard information
| GHS classification*: serious eye damage/eye irritation, category 2A | |
| H319—Causes serious eye irritation | |
| GHS classification: hazardous to the aquatic environment, long-term hazard, category 2 | |
| H411—Toxic to aquatic life with long-lasting effects | |
**Globally Harmonized System of Classification and Labeling of Chemicals. Explanation of pictograms.
Zinc dibutyl dithiophosphate fast facts
| CAS Reg. No. | 6990-43-8 |
| Empirical formula | C16H36O4P2S4Zn |
| Molar mass | 548.07 g/mol |
| Appearance | Oily solid or liquid |
| Melting point | 36.8 ºC |
| Boiling point | 303.7 ºC |
| Water solubility | Insoluble |
| CAS Reg. No. | 1306278-47-6 |
| SciFinder nomenclature | Carbonimidic diazide, N-(5-azido-1H-tetrazol-1-yl)- |
| Empirical formula | Empirical formula |
| Molar mass | 220.12 g/mol |
| Appearance | Red crystals |
| Melting point | 78 ºC |
| Water solubility | Dec. to HN3 |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

