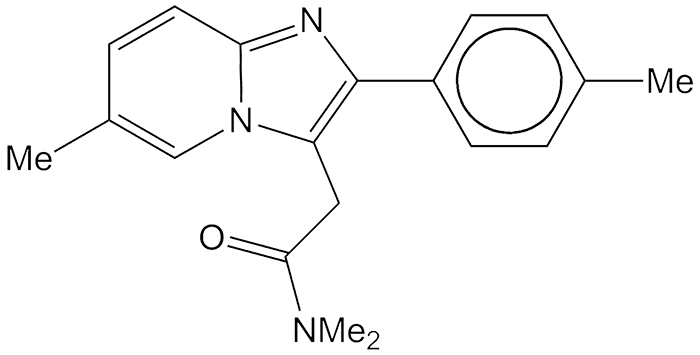
Zolpidem—sold under a host of brand names, the best known of which is Ambien—is a medication used to treat insomnia. The article of commerce is the tartrate salt. In most people, it induces sleep rapidly and is effective for ≈2–3 hours.
The US patent on zolpidem was issued in 1984 to the French company Synthélabo, now part of Sanofi-Aventis. By 2007, it was off-patent; and the US Federal Drug Administration approved 13 generic versions of zolpidem tartrate.
Early in 2015, M. J. Miller and colleagues at the University of Notre Dame investigated whether zolpidem could be used to treat tuberculosis. They found that zolpidem has some activity against Mycobacterium tuberculosis replication, but some of its structural isomers have much greater potency. The authors believe that "further optimization of the [zolpidem] core may well 'put TB to rest'."

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

