FOR IMMEDIATE RELEASE
ACS News Service Weekly PressPac: April 27, 2011
A better imaging agent for heart disease and breast cancer
“Large-Scale Synthesis of Bioinert Tantalum Oxide Nanoparticles for X-ray Computed Tomography Imaging and Bimodal Image-Guided Sentinel Lymph Node Mapping”
Journal of the American Chemical Society
Scientists are reporting development of a process for producing large quantities of a much-needed new imaging agent for computed tomography (CT) scans in heart disease, breast cancer and other diseases, and the first evidence that the material is safe for clinical use. The imaging agent is a tantalum oxide nanoparticle, which is inexpensive, and stays in the body long enough to image many different organs. The report appears in the Journal of the American Chemical Society.
Taeghwan Hyeon, Seung Hong Choi, and colleagues explain that CT is one of the most widely used medical imaging tests and highly effective in diagnosing disease. However, CT requires use of large amounts of imaging agents, which make body structures more visible. Today’s imaging agents have many disadvantages. They don’t stay in the body for very long, for instance, limiting the types of images that physicians can obtain. Nanoparticles have been developed as imaging agents, but they have been made of either gold (very expensive) or bismuth (toxic). So, the researchers wanted to develop a new nanoparticle imaging agent that would overcome those disadvantages.
The researchers describe development and successful initial tests of large batches of tantalum oxide nanoparticles that exhibited “remarkable performances” in imaging tests of the heart, lymph nodes, kidneys, and other structures in laboratory rats, which are stand-ins for humans in such research. Regarding safety, they say, “The nanoparticles did not affect normal functioning of organs.” The particles could have especially important uses in checking blood vessels in the heart for blockages, and lymph nodes for the spread of breast cancer, the report states.
The authors acknowledge funding from the Korean Ministry of Education, Science, and Technology, as well as the National Research Foundation of Korea.
![]()
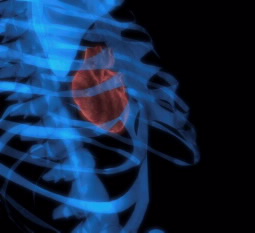
nosis of heart disease, breast cancer, and other
conditions.
Contact
Science Inquiries: Michael Woods, Editor, 202-872-6293
General Inquiries: Michael Bernstein, 202-872-6042


