What molecule am I?
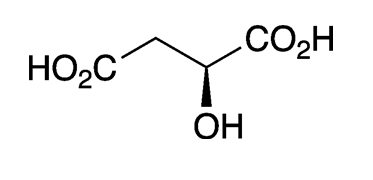
Malic acid, a hydroxydicarboxylic acid, is found in all forms of life. It exists naturally only as the L-enantiomer. It should not be confused with the similar sounding maleic and malonic acids.
L-Malic acid gives many fruits, particularly apples, their characteristic flavor. It is often referred to as “apple acid”. The word malic is derived from the Latin mālum, for which Malus, the genus that contains all apple species, is also named.
The global market size for malic acid (natural and manufactured1) is ≈US$200 million; the US market is ≈$35 million. The primary end use in the United States is for flavoring beverages, foods, and confectionaries, with much smaller quantities used in cosmetics and personal care products. The price of malic acid ranges from US$0.90 to $10.00/kg, depending on the purity, quantity, and end use.
1. The manufactured product is racemic.
L-Malic acid hazard information*
| Hazard class** | Hazard statement | |
|---|---|---|
| Skin corrosion/irritation, category 2 | H315—Causes skin irritation | |
| Serious eye damage/eye irritation, category 2A | H319—Causes serious eye irritation | |
| Specific target organ toxicity, single exposure, respiratory tract irritation, category 3 | H335—May cause respiratory irritation | |
* Compilation of three safety data sheets.
** Globally Harmonized System of Classification and Labeling of Chemicals.
Explanation of pictograms.
This molecule was suggested by a reader. We present almost all of the molecules suggested by our readers. If you have a molecule you would like us to consider, please send us a message. And thank you for your interest in Molecule of the Week! —Ed.
L-Malic acid fast facts
| CAS Reg. No. | 97-67-6 |
| SciFinder nomenclature | Butanedioic acid, 2-hydroxy-, (2S)- |
| Empirical formula | C4H6O5 |
| Molar mass | 134.09 g/mol |
| Appearance | White crystalline powder, granules, or needles |
| Melting Point | 130 ºC |
| Water solubility | 558 g/L |

Learn more about this molecule from CAS, the most authoritative and comprehensive source for chemical information.
Molecule of the Week needs your suggestions!
If your favorite molecule is not in our archive, please send us a message. The molecule can be notable for its current or historical importance or for any quirky reason. Thank you!
Stay Ahead of the Chemistry Curve
Learn how ACS can help you stay ahead in the world of chemistry.

